Baumgartner H, De Backer J, Babu-Narayan SV, et al; ESC Scientific Document Group. 2020 ESC Guidelines for the management of adult congenital heart disease. Eur Heart J. 2021 Feb 11;42(6):563-645. doi: 10.1093/eurheartj/ehaa554. PMID: 32860028.
Stout KK, Daniels CJ, Aboulhosn JA, et al. 2018 AHA/ACC Guideline for the Management of Adults With Congenital Heart Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019 Apr 2;139(14):e637-e697. doi:10.1161/CIR.0000000000000602. PMID: 30586768.
Definition, Etiology, Pathogenesis Top
Eisenmenger syndrome (pulmonary vascular disease leading to shunt reversal) is a result of an untreated large left-to-right shunt (interatrial, interventricular, or aortopulmonary) and increased pulmonary perfusion, leading to severe irreversible pulmonary hypertension and shunt reversal (ie, a right-to-left shunt). It is a complication of simple or complex defects associated with large shunts. Eisenmenger syndrome frequently develops in childhood; in adults, the diagnosis is made mainly in patients with an atrioventricular septal defect, large interatrial shunt, nonrestrictive ventricular septal defects, or patent ductus arteriosus (PDA).
Clinical Features Top
1. Symptoms: Significant reduction in exercise tolerance, resting dyspnea exacerbated with exercise, palpitations, and chest pain. Patients with high hematocrit levels develop symptoms of hyperviscosity and hemoptysis. Syncope may occur in patients with advanced disease. Cyanosis and symptoms are often exacerbated in conditions associated with decreased systemic vascular resistance. This typically happens during fever.
2. Signs: Central cyanosis, clubbing of the nail beds, evidence of right ventricular lift, and a palpable second heart sound. Auscultation often reveals accentuation of the second heart sound; a right ventricular third heart sound and fourth heart sound may be present in patients with right ventricular hypertrophy. Disappearance of the murmurs caused by the underlying defect, for instance, the continuous murmur caused by PDA or the systolic murmur caused by a ventricular septal defect, should raise the suspicion of development of Eisenmenger physiology. Frequently, a soft systolic murmur at the left sternal border is audible, which is probably due to the dilation of the main pulmonary artery. In patients with pulmonary regurgitation, a relatively low-grade proto-meso-diastolic murmur, resulting from a high pulmonary vascular resistance (Graham Steell murmur), may be audible. In individuals with Eisenmenger PDA differential cyanosis is seen (cyanosis and clubbing in the lower extremities only).
3. Natural history: Patients with Eisenmenger syndrome and simple defects (shunts) have an average life expectancy >40 years, while in those with complex defects, the survival is shorter. The most common causes of death are heart failure, sudden cardiac death, and massive hemoptysis. The risk factors for severe complications or death include pregnancy, general anesthesia, dehydration, hemorrhage, surgical procedures (noncardiac), diuretics overuse, certain oral contraceptives, anemia (usually after unnecessary phlebotomies), cardiac catheterization, intravenous therapy, and pulmonary infections.
Diagnosis Top
Diagnosis is based on the presence of a high pulmonary vascular resistance that does not respond to vasodilators (oxygen, nitric oxide) in a patient with a congenital heart disease with a shunt. Invasive diagnostic methods are indicated when noninvasive assessment suggests feasibility of surgical repair of the defect.
1. Laboratory tests reveal abnormalities that are secondary to hypoxemia; hemoglobin oxygen saturation (SaO2) is usually <90% (measured at rest for at least 5 minutes). Complete blood count (CBC), hematocrit, renal function assessment, clotting profile, and serum uric acid levels should be measured and could be used for longitudinal monitoring. Further testing with additional tests to assess iron indices, serum brain natriuretic peptide, folic acid, and vitamin B12 can be done.
2. Electrocardiography (ECG): Features of right atrial enlargement (if in sinus rhythm) and features of right ventricular hypertrophy (often with strain pattern) and overload are the dominant features of ECG.
3. Chest radiography (Figure 1): The size of the heart may vary depending on the primary defect and severity of Eisenmenger syndrome. Pulmonary perfusion is clearly reduced, with a pruning pattern of the arteries; the hilar vessels are dilated.
4. Echocardiography: Right ventricular hypertrophy with color and spectral Doppler evidence of bidirectional flow through the shunt, and severe elevation in right ventricular systolic pressures. Often, there is an associated significant dilation of pulmonary arteries. Other abnormalities such as mitral or tricuspid regurgitation can be present and depend on the severity of Eisenmenger syndrome. Transesophageal echocardiography (TEE), magnetic resonance imaging (MRI), or computed tomography (CT) may detect proximal pulmonary artery thrombosis.
5. Exercise capacity should be assessed with the 6-minute walk test.
Treatment Top
1. General measures:
1) Administration of influenza vaccine every year, and pneumococcal vaccine every 5 years.
2) Monitoring for the signs and symptoms of bleeding.
3) Aggressive and prompt treatment of upper respiratory tract infections.
4) Avoidance of dehydration, excessive physical exercise, and staying at high altitudes. Smoking is prohibited.
5) Patients should be followed in specialized centers with expertise in care of adults with congenital heart disease and pulmonary hypertension.
6) Contraceptive advice: Pregnancy is a contraindication in women with Eisenmenger syndrome. Given the high failure rates, the use of single-barrier methods is not recommended. Estrogen-containing contraceptives should be avoided in these patients.
2. Treatment of hyperviscosity should only be performed in patients with hematocrit >65% who have moderate to severe symptoms of hyperviscosity and in whom iron deficiency and dehydration have been excluded. Perform therapeutic phlebotomy of 250 to 500 mL of blood with isovolumic fluid replacement. An air filter is recommended for all intravenous lines to avoid air embolism.
3. Iron supplementation should be performed in all patients with iron deficiency anemia related to repeated phlebotomies.
4. Treatment of arrhythmias: Sinus rhythm should be maintained with individualization of antiarrhythmic therapy. Transvenous leads must be avoided due to the increased risk of paradoxical embolization.
5. Treatment of hemoptysis is usually not necessary; however, an initial assessment with chest radiography followed by CT imaging of the chest if an infiltrate is present is recommended. Life-threatening events (rupture of an aortopulmonary collateral artery, pulmonary artery, or arteriole) are an indication for percutaneous embolization or surgery. Bronchoscopy should be avoided in patients with Eisenmenger syndrome and hemoptysis.
6. Targeted treatment of pulmonary hypertension: In patients with World Health Organization (WHO) functional class III symptoms, the endothelin-receptor antagonist (ERA) bosentan should be initiated.Evidence 1Strong recommendation (benefits clearly outweigh downsides; right action for all or almost all patients). Moderate Quality of Evidence (moderate confidence that we know true effects of the intervention). Quality of Evidence lowered due to the combination of a short trial duration, small number of patients, and lack of data on longer-term consequences of use in terms of patient-important outcomes. Galie N, Beghetti M, Gatzoulis MA, Granton J, Berger RM, Lauer A, Chiossi E, Landzberg M; Bosentan Randomized Trial of Endothelin Antagonist Therapy-5 (BREATHE-5) Investigators. Bosentan therapy in patients with Eisenmenger syndrome: a multicenter, double-blind, randomized, placebo-controlled study. Circulation. 2006 Jul 4;114(1):48-54. Epub 2006 Jun 26. PubMed PMID: 16801459. Other ERAs, phosphodiesterase inhibitors (eg, sildenafil, tadalafil), and prostacyclins can also be considered in patients with class III symptoms. In selected patients combination therapy may be considered. Also see Pulmonary Hypertension. Initiation of therapy and follow-up is recommended to take place in conjunction with specialized pulmonary hypertension clinics.
7. Single-lung or total heart-lung transplant may be used in patients with severe hypoxemia or heart failure and no contraindications to surgery in whom the risk of death within 1 year is >50%. Single-lung transplant is performed in conjunction with repair of the cardiac defect (eg, ventricular septal defect) in selected cases.
8. Treatment of hyperuricemia: Asymptomatic hyperuricemia does not need treatment. In patients with acute gout, treatment with colchicine and anti-inflammatory drugs should be instituted with attention to the increased risk of renal function deterioration and increased risk of bleeding. Also see Gout.
9. Noncardiac surgery: This should only be performed when absolutely necessary and unavoidable due to the high mortality associated with Eisenmenger syndrome. An experienced cardiac anesthesiologist with a clear understanding of Eisenmenger physiology should be requested to administer anesthesia. An air filter should be used on all intravenous lines. Preoperative phlebotomy (with isovolumic fluid replacement) can be considered in patients with hematocrit >65% (in absence of hyperviscosity symptoms). The blood withdrawn can be used for autologous transfusion if needed.
10. Endocarditis prophylaxis is indicated in all patients with cyanotic congenital heart disease.Evidence 2Strong recommendation (benefits clearly outweigh downsides; right action for all or almost all patients). Low Quality of Evidence (low confidence that we know true effects of the intervention). Quality of Evidence lowered due to the lack of experimental data. Wilson W, Taubert KA, Gewitz M, et al; American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee; American Heart Association Council on Cardiovascular Disease in the Young; American Heart Association Council on Clinical Cardiology; American Heart Association Council on Cardiovascular Surgery and Anesthesia; Quality of Care and Outcomes Research Interdisciplinary Working Group. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007 Oct 9;116(15):1736-54. Epub 2007 Apr 19. Erratum in: Circulation. 2007 Oct 9;116(15):e376-7. PubMed PMID: 17446442.
11. Routine anticoagulation: Although patients with Eisenmenger syndrome are at risk of thrombosis (especially in the dilated pulmonary arteries), currently available data do not support the routine use of anticoagulation in these patients due to the inherent increased risk of bleeding as well as difficulty with the routine monitoring of anticoagulation.
FiguresTop
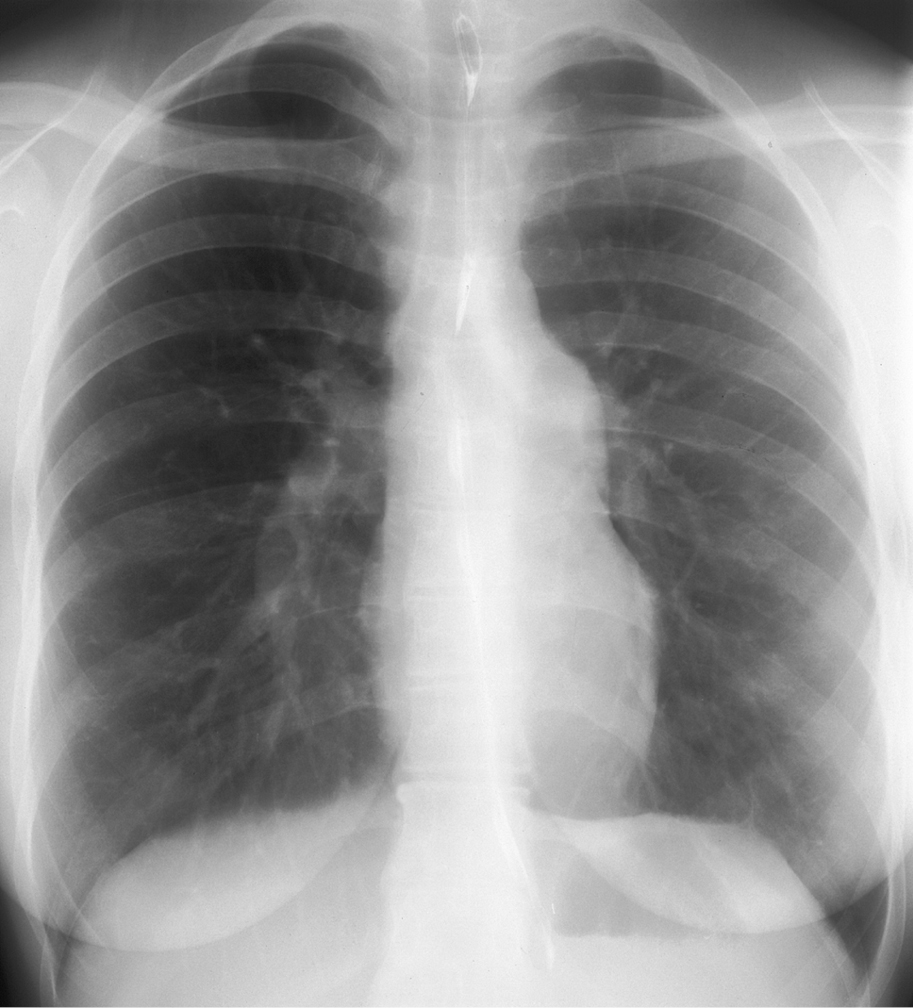
Figure 3.7-1. Chest radiography of a patient with Eisenmenger syndrome: a normal-sized cardiac silhouette, prominent main pulmonary artery, enlarged pulmonary arteries, diminished peripheral vascular markings.