Etiology and PathogenesisTop
1. Etiologic agent: Four serotypes of the dengue virus (DENV 1-4) of the genus Flavivirus, family Flaviviridae. Past infection confers immunity to the specific DENV serotype but does not protect against infection with other serotypes.
2. Pathogenesis: The pathophysiologic mechanisms responsible for manifestations of dengue are complex and not fully known. The release of cytokines and other proinflammatory and vasoactive factors by immune cells (eg, monocytes, T helper cells, cytotoxic T cells, mast cells) and platelet stimulation in response to DENV infection lead to endothelial activation and damage as well as to coagulation and fibrinolysis disorders, which are particularly pronounced in severe disease. The key mechanism responsible for severe disease is increased plasma leakage. Second or subsequent new infection with a different DENV serotype may be associated with increased viral uptake by Fcy receptors of immune cells and enhanced replication of the pathogen. This is caused by bloodstream presence of antibodies produced during the previous infection, which are unable to effectively neutralize the heterologous virus. This mechanism of antibody-dependent enhancement of infection may lead to extensive release of proinflammatory cytokines and is associated with increased risk of severe secondary infection.
3. Reservoir and transmission: The reservoir for dengue is humans and, in some regions, monkeys. The vector is Aedes genus mosquitoes, of which the most important is A aegypti. Dengue is not transmitted from person to person through direct contact; however, rare cases of infection through exposure to blood of an infected person in the viremic phase (laboratory accidents, needlestick injuries), blood transfusion, or bone marrow or organ transplant have been documented. Also, 2 cases of transmission through sexual contact have been reported to date. Vertical mother-to-child transmission in the perinatal period and transmission through breastfeeding are possible.
4. Risk factors for infection: Staying within areas with active DENV transmission. Risk factors for severe dengue: DENV-2 infection, history of infection with a different DENV serotype (postulated mechanism of antibody-dependent enhancement of infection), genetic predisposition, age <11 years.
5. Incubation and contagious period: The average incubation period is 5 to 7 days (sometimes 3-10 days). In the viremic phase (starting several hours before symptom onset until defervescence, usually ≤5 days), the patient may transmit the infection onto mosquitoes.
EpidemiologyTop
Dengue is endemic to numerous tropical and subtropical regions, including South and Southeast Asia, the Caribbean, South and Middle Americas, Pacific islands, and selected African countries (www.cdc.gov). The estimated incidence of DENV infections is 390 million cases per year, with the majority occurring in Asia. Local environmental conditions, including, in particular, air temperature and precipitation, have major influence on the life cycle and breeding of mosquitoes that serve as DENV vectors and the intensity of transmission. In recent years DENV infections have also been observed in the south of Europe (Croatia, France, Spain, Italy); a large outbreak was reported on Madera (Portugal) in 2012. In 2019, 4020 cases were confirmed in the European Union/European Economic Area. Nearly all of those occurred in travelers, usually those returning from Thailand or India.
clinical features and natural historyTop
In 75% of infected individuals the disease is asymptomatic, while 5% develop severe dengue.
The disease has several phases (Figure 10.11-1). Presenting signs and symptoms in the febrile phase include high-grade fever, severe headache with characteristic retro-orbital pain, nausea, muscle pain, arthralgia, and weakness. Some patients develop rash, most frequently a blanchable maculopapular rash during the first 24 to 48 hours of fever, followed by a convalescent confluent petechial rash 2 to 3 days after defervescence. A pathognomonic sign of dengue is a petechial rash on extremities, with well-demarcated noninvolved skin, otherwise called “white islands in a sea of red” (Figure 10.11-2). Some patients have symptoms of pharyngitis early during the disease. Liver injury and jaundice may also develop.
After a few days the fever resolves and the majority of patients improve. Loss of appetite (or even anorexia) and malaise may persist for much longer. In severe dengue at the time when fever decreases to 37.5 to 38 degrees Celsius (critical phase, usually 24-48 h from fever resolution, lasting ≤48 h), the patient’s condition deteriorates rapidly. Symptoms of increased plasma leakage manifest in this phase, including plasma leakage into body cavities (particularly pleural and peritoneal cavities), sometimes with drop in blood pressure, loss of consciousness, and shock. Disseminated intravascular coagulation and hypoalbuminemia may also develop. The risk of severe dengue is increased in patients with warning signs (Table 10.11-1).
Mucosal bleeding of varying intensity is a typical manifestation of dengue. In severe disease patients may have massive gastrointestinal or urogenital bleeding. The risk of clinically significant bleeding increases in patients with shock, kidney or liver failure, metabolic acidosis, those treated with nonsteroidal anti-inflammatory drugs (NSAIDs) or anticoagulants, those with peptic ulcer disease, and those with any tissue damage, including post IM injections.
Rarely severe dengue may manifest with acute hepatitis, myocarditis, pancreatitis, and/or encephalitis.
Convalescent phase: Absorption of the extravasated fluids and hemodynamic stabilization. Improper fluid resuscitation may result in fluid overload and pulmonary edema.
DIAGNOSISTop
1. Identification of the etiologic agent:
1) Molecular tests in the viremic phase: Real-time polymerase chain reaction (RT-PCR), sample material: blood.
2) Isolation of the virus from blood in the viremic phase.
3) Serologic tests: The presence of IgM antibodies indicates recent DENV infection, whereas detection of IgG antibodies in a single sample confirms past infection. Sometimes in secondary infection (infection with a different serotype, usually after ≥3-6 months from the primary infection), due to some cross-immunity between serotypes, the IgM antibody response is poor or delayed. In these cases seroconversion or a 4-fold increase in IgG titers in paired acute and convalescent sera samples indicate recent infection.
4) Detection of DENV antigen: Rapid immunochromatographic tests and enzyme-linked immunosorbent assay (ELISA) that detect dengue nonstructural protein-1 (NS-1) antigen in serum are useful as auxiliary studies.
Utility of laboratory tests in the diagnostic workup of dengue: Figure 10.11-3.
2. Other tests: Basic laboratory blood tests usually show leukopenia and minor thrombocytopenia. More severely ill patients may have elevated hematocrit (Ht) (warning sign) and serum alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels.
The following clinical classification of dengue was proposed by the World Health Organization (WHO) in 2009: Probable dengue, laboratory-confirmed dengue (with or without warning signs), and severe dengue (Table 10.11-1).
Other viral hemorrhagic fevers, rickettsial diseases, malaria, influenza, HIV infection (acute retroviral syndrome), chikungunya fever, measles, mononucleosis, acute pharyngitis, enteric fever, paratyphoid fevers, leptospirosis.
TREATMENTTop
No antiviral treatment is available.
In mild dengue use:
1) Oral rehydration.
2) Antipyretic and analgesic treatment: Acetaminophen (INN: paracetamol) and nonpharmacologic fever management methods.
Avoid acetylsalicylic acid (ASA) and other NSAIDs that may worsen bleeding. Monitor the patient for progression to severe disease (complete blood count, warning signs, progression to critical phase after fever resolution). The presence of warning signs is associated with high risk of life-threatening complications and is an indication for hospitalization and careful monitoring of the patient (sometimes in intensive care units [ICUs]). Vigorous fluid resuscitation is crucial, as it reduces mortality in patients with increased plasma leakage. For fluid resuscitation use 0.9% NaCl or Ringer lactate; to optimize treatment assess Ht and clinical condition of the patient several times a day. In patients with shock that does not respond to crystalloids use colloids.
Transfusion of platelet concentrates in patients with thrombocytopenia and routine use of glucocorticoids are not recommended. Severe bleeding or decreased Ht levels in patients with unstable vital signs are indications for packed red blood cell (PRBC) or whole blood transfusions. When making a decision on PRBC transfusion, do not use the Ht criterion of <30%, as patients with severe dengue usually have elevated Ht at baseline due to increased plasma leakage.
special considerationsTop
Data on the effects of dengue in pregnancy on fetal development are limited. Severe dengue in pregnancy is associated with increased risk of premature birth and low birth weight of the newborn.
preventionTop
Vaccination: Live attenuated tetravalent vaccine against yellow fever and dengue (CYD-TDV) is approved, among others, in Brazil, Mexico, El Salvador, Paraguay, Costa Rica, Indonesia, the Philippines, and the United States (for residents of dengue-endemic regions). The WHO recommends it in individuals aged 9 to 45 years with a history of DENV infection confirmed by serologic tests (paradoxically, vaccination may increase the risk of severe dengue infection in a person without previous infection [seronegative at the time of vaccination]). Therefore, vaccination is not recommended in the majority of travelers to tropics.
1. Mosquito bite precautions.
2. Preventing vector breeding and dissemination in regions with active DENV transmission: Eradication of mosquito breeding grounds (eg, removal of artificial fresh water reservoirs, tight covering of containers with water), use of insect repellents. A release of A aegypti infected with Wolbachia pipientis (an intracellular bacterium that reduces the insects’ susceptibility to DENV infection) into dengue-endemic areas is associated with reduced incidence of symptomatic DENV infections and hospitalizations due to dengue among residents of such regions.
3. Patient isolation: Not needed. In regions with favorable conditions for DENV transmission, patients with viremia should stay in a place free of mosquitoes or use mosquito repellents to avoid being bitten.
TABLES AND FIGURESTop
|
Probable dengue |
|
Staying in dengue-endemic area. Fever and ≥2 of the following criteria: 1) Nausea, vomiting 2) Rash 3) Headache and/or arthralgia 4) Positive tourniquet test result 5) Leukopenia 6) Any warning signa |
|
Laboratory-confirmed dengue |
|
Severe dengue |
|
1) Severe plasma leakage (shock, respiratory distress) 2) Severe bleeding (as evaluated by clinician) 3) Severe organ dysfunction (eg, liver injury with ALT or AST ≥1000 U/L, altered mental status) |
|
a Warning signs: Abdominal pain or tenderness, persistent vomiting, clinical fluid accumulation (ascites, pleural effusion), mucosal bleeding, lethargy or restlessness, hepatomegaly, elevated Ht with a decrease in platelet count. |
|
Adapted from World Health Organization. Dengue Guidelines for the Diagnosis, Treatment, Prevention and Control. Edition 2009. |
|
ALT, alanine aminotransferase; AST, aspartate aminotransferase; Ht, hematocrit; WHO, World Health Organization. |
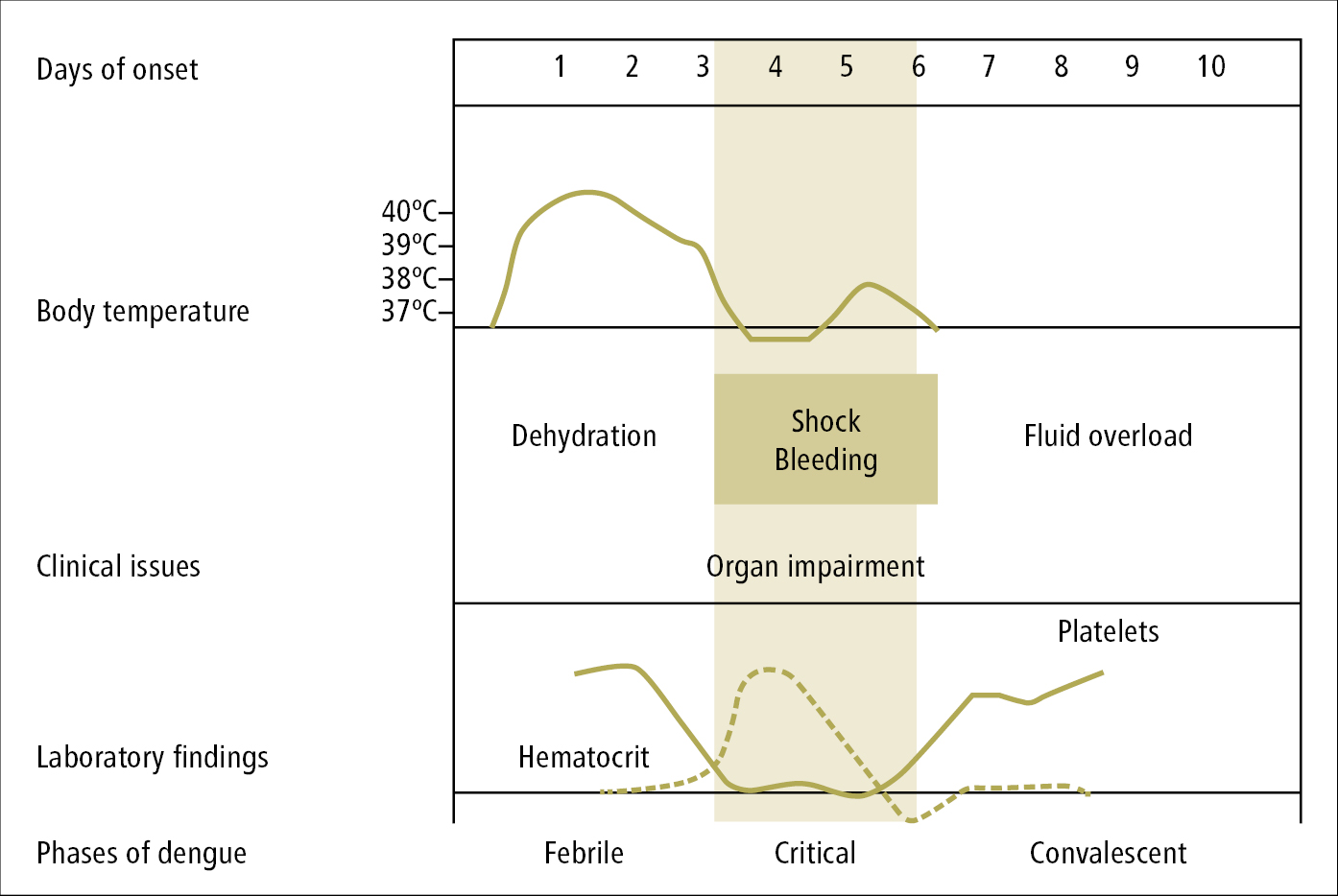
Figure 10.11-1. Dengue phases, signs, symptoms, and CBC findings according to the WHO (2009).
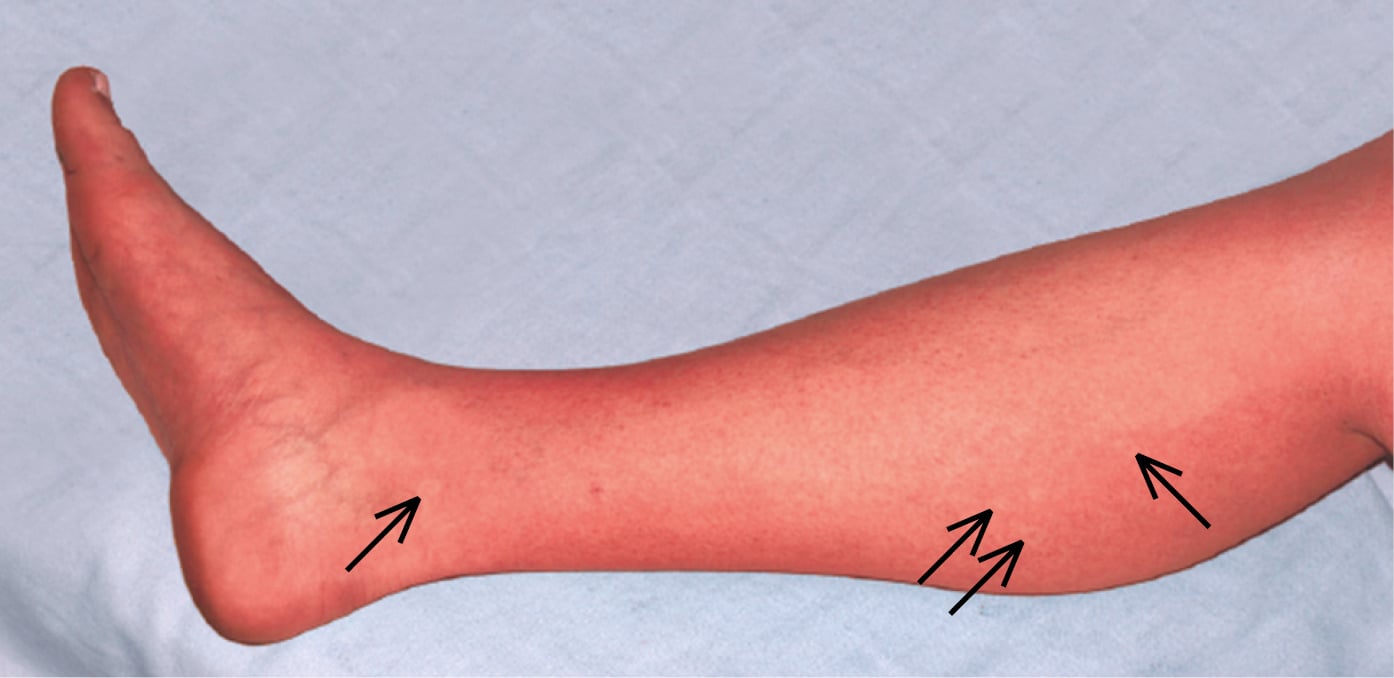
Figure 10.11-2. Dengue rash with visible “white islands in a sea of red.”
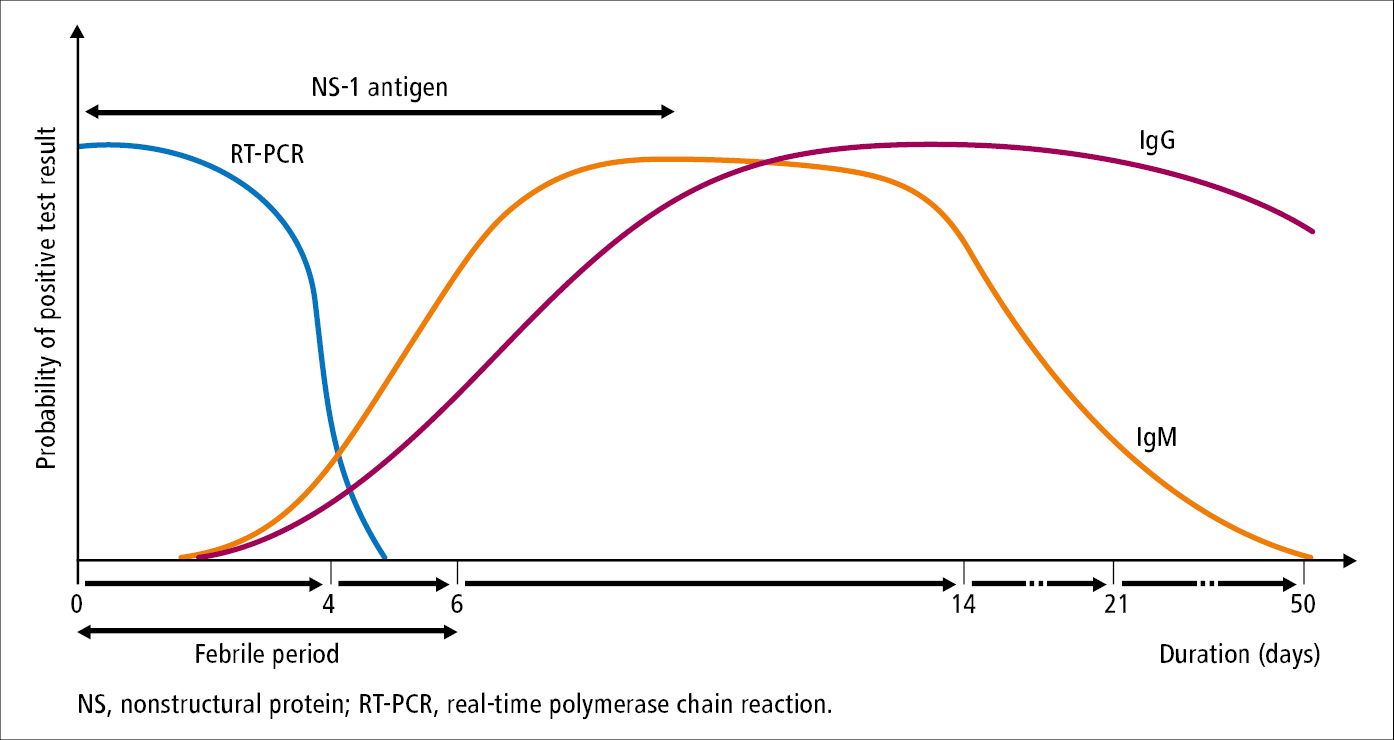
Figure 10.11-3. Utility of laboratory tests in the diagnostic workup of dengue.