Xu G, Walker DH, Jupiter D, Melby PC, Arcari CM. A review of the global epidemiology of scrub typhus. PLoS Negl Trop Dis. 2017 Nov 3;11(11):e0006062. doi: 10.1371/journal.pntd.0006062. PMID: 29099844; PMCID: PMC5687757.
Rajapakse S, Weeratunga P, Sivayoganathan S, Fernando SD. Clinical manifestations of scrub typhus. Trans R Soc Trop Med Hyg. 2017 Feb 1;111(2):43-54. doi: 10.1093/trstmh/trx017. PMID: 28449088.
Janardhanan J, Trowbridge P, Varghese GM. Diagnosis of scrub typhus. Expert Rev Anti Infect Ther. 2014 Dec;12(12):1533-40. doi: 10.1586/14787210.2014.974559. Epub 2014 Oct 31. PMID: 25359599.
Etiology and PathogenesisTop
1. Etiologic agent: Orientia tsutsugamushi rickettsia, an obligate intracellular gram-negative bacterium transmitted by infected chiggers (larval mites). Scrub typhus is prevalent in areas dominated by vectors in the mite egg-laying season (in the tropics this is most often during the wet season): Northern Australia, Oceania, South Asia, and Central East Asia (from Russian Far East and Japan to Afghanistan). The infection frequency is high; in some Southeast Asian countries, ~3% of the population acquires infection every month.
2. Pathogenesis: O tsutsugamushi infects endothelial cells in small veins, arteries, and capillaries. Subsequent cell damage leads to vasculitis and formation of small thrombi. Loss of epithelial integrity and transudates causing hypovolemia and decreased tissue perfusion may also occur. The disease affects multiple organs simultaneously and may result in their failure.
3. Transmission route: Bite of an infected chigger (Leptotrombidium) inhabiting areas of heavy scrub vegetation.
4. Incubation period: 1 to 2 weeks (6-21 days) on average.
Clinical Features Top
Signs and symptoms are nonspecific (fever, headache, myalgia). Cough, nausea, vomiting, and abdominal pain may also occur. A painful eschar (initially presenting as a small papule, subsequently enlarging and accompanied by central necrosis, resembling a cigarette burn) may develop at the bite site in prefebrile phase (Figure 10.3-1). Regional lymph nodes may be enlarged. Generalized lymphadenopathy is sometimes observed. Towards the end of the first week of infection, ~50% of infected individuals have macular or maculopapular rash, which starts on the trunk and spreads to the extremities. Systemic involvement, if present, is observed at the beginning of the second week of infection:
1) Cardiovascular system: Myocarditis and pericarditis, arrhythmia.
2) Respiratory system: Pneumonia.
3) Nervous system: Meningitis, encephalitis, cranial nerve palsy, cerebellar inflammation, polyneuropathy, transverse myelitis.
4) Digestive system: Diarrhea, pancreatitis, cholecystitis, gastrointestinal bleeding.
5) Urinary system: Acute renal failure, nephrotic syndrome.
6) Hematopoietic system: Disseminated intravascular coagulation (DIC), hemophagocytic syndrome, thrombocytopenia, splenomegaly.
7) Other: Some patients have conjunctival congestion. Severe disease may result in multiorgan failure associated with high risk of death.
DiagnosisTop
1. Identification of the etiologic agent:
1) Culture is challenging and performed only by specialized biosafety level 3 (BSL3) laboratories (material: whole blood, skin biopsy specimen obtained from the eschar or area affected by rash [>4 mm; higher sensitivity than with blood], unpreserved autopsy material); culture has the highest sensitivity in the acute phase, before the initiation of antibacterial treatment.
2) Molecular tests: Detection of the rickettsial DNA using polymerase chain reaction (PCR) and quantitative PCR (qPCR) (material: whole blood, serum [lower sensitivity than with blood], skin biopsy specimen obtained from the eschar or area affected by rash, eschar scrapings/swab [lower sensitivity than with biopsy], formalin-fixed or unpreserved autopsy material [low sensitivity]). Molecular tests have the highest sensitivity in the first weeks of infection, up to 48 hours from antibacterial treatment initiation.
3) Serologic testing is a basic diagnostic method that enables detection of IgG and IgM antibodies (indirect immunofluorescence or enzyme-linked immunosorbent assay [ELISA] are preferred). A 4-fold titer increase within several weeks or seroconversion confirm the diagnosis (repeat testing after 2-4 weeks of disease resolution). IgM antibodies appear in the first week of infection, and IgG antibodies, at the end of the second week. Cross-reactions between different rickettsiae are possible. The risk of obtaining false-positive results is higher in patients with infectious mononucleosis and HIV infection. Antibodies against rickettsiae are present in serum for years after infection resolution, which is why antibody testing is not used for monitoring of treatment efficacy. The Weil-Felix reaction, which is a heterophile antibody test, is commonly used in resource-limited settings but has low sensitivity and specificity.
2. Other:
1) Laboratory tests: Leukocyte count may be increased or normal; thrombocytopenia, increased aminotransferase activity, and increased creatinine levels are often observed.
2) Imaging: Chest radiography or computed tomography (CT) may reveal ground-glass opacities and consolidations.
Malaria, dengue, Japanese encephalitis, other rickettsial diseases, leptospirosis, enteric fever, cutaneous anthrax.
TreatmentTop
Oral or IV doxycycline 100 mg every 12 hours for 7 days; if contraindicated, oral or IV azithromycin 500 mg in a single dose for 5 days or oral or IV chloramphenicol 500 mg every 6 hours for 7 days.
PrognosisTop
Mortality among treated individuals is 1.4%, whereas among those untreated, ~30% (median, 6%; the risk increases with age).
PreventionTop
Vaccination: None available.
Using insect repellents (eg, N,N-diethyl-meta-toluamide [DEET]) or insecticides (eg, permethrin): see Infection Prevention: Nonspecific Insect and Tick Bite Precautions.
FiguresTop
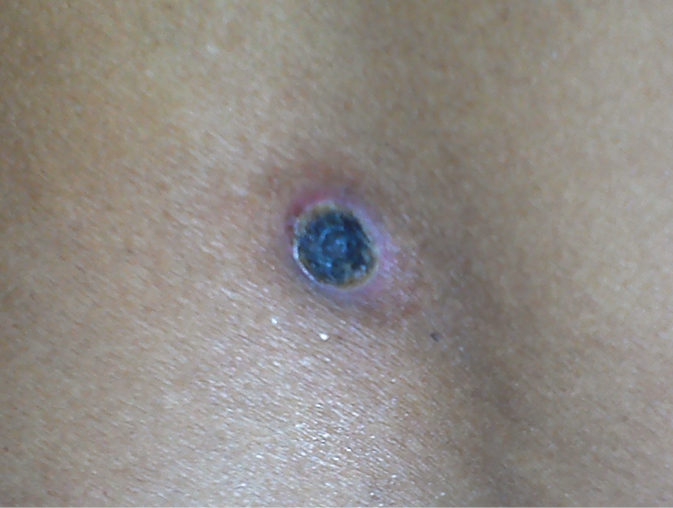
Figure 10.3-1. Eschar at the site of a chigger bite in scrub typhus. Photograph courtesy of Dr Anupam Prakash.