Rabi DM, McBrien KA, Sapir-Pichhadze R, et al. Hypertension Canada's 2020 Comprehensive Guidelines for the Prevention, Diagnosis, Risk Assessment, and Treatment of Hypertension in Adults and Children. Can J Cardiol. 2020 May;36(5):596-624. doi: 10.1016/j.cjca.2020.02.086. PMID: 32389335.
Butalia S, Audibert F, Côté AM, et al; Hypertension Canada. Hypertension Canada's 2018 Guidelines for the Management of Hypertension in Pregnancy. Can J Cardiol. 2018 May;34(5):526-531. doi: 10.1016/j.cjca.2018.02.021. Epub 2018 Mar 1. PMID: 29731014.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2018 Oct 23; 138(17): e426-e483. doi: 10.1161/CIR.0000000000000597. PMID: 30354655.
Williams B, Mancia G, Spiering W, Agabiti Rosel E, et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J. 2018 Sep 1; 29(33):3021-3104. doi: 10.1093/eurheartj/ehy339. PMID: 30165516.
Persu A, Giavarini A, Touzé E, et al; ESH Working Group Hypertension and the Kidney. European consensus on the diagnosis and management of fibromuscular dysplasia. J Hypertens. 2014 Jul;32(7):1367-78. doi: 10.1097/HJH.0000000000000213. Review. PMID: 24842696.
European Society of Gynecology (ESG); Association for European Paediatric Cardiology (AEPC); German Society for Gender Medicine (DGesGM), Regitz-Zagrosek V, Blomstrom Lundqvist C, Borghi C, et al; ESC Committee for Practice Guidelines. ESC Guidelines on the management of cardiovascular diseases during pregnancy: the Task Force on the Management of Cardiovascular Diseases during Pregnancy of the European Society of Cardiology (ESC). Eur Heart J. 2011 Dec;32(24):3147-97. doi: 10.1093/eurheartj/ehr218. PMID: 21873418.
National Institute for Health and Care Excellence. Hypertension in pregnancy: diagnosis and management. https://www.nice.org.uk/guidance/cg107. Published August 2010. Updated January 2011. Accessed March 22, 2017.
Definition, Etiology, PathogenesisTop
Renovascular hypertension is a secondary cause of hypertension due to renal ischemia and subsequent hypersecretion of renin. Ischemic nephropathy is a reduction in the glomerular filtration rate (GFR) and impairment of other kidney functions due to hemodynamically significant renal artery stenosis.
Causes of renal artery stenosis: The most common causes are atherosclerosis (usual risk factors apply) and fibromuscular dysplasia. Other causes include renal artery aneurysm, arterial embolus, arteriovenous fistula (congenital/traumatic), segmental arterial occlusion (posttraumatic), extrinsic, compression of renal artery (eg, pheochromocytoma), renal compression (eg, metastatic tumor), stenosis to a solitary functioning kidney, aortic coarctation, systemic vasculitis (eg, Takayasu, polyarteritis), atheroembolic disease, vascular occlusion due to an endovascular aortic stent graft.
A hemodynamically significant stenosis (ie, >60%-70% of the artery diameter) of the renal artery or arteries results in hypoperfusion of the kidney as well as an increase in the activity of the renin-angiotensin-aldosterone system (RAAS) and its subsequent effects.
Clinical Features and Natural HistoryTop
1. Clinical features of renovascular hypertension:
1) A sudden onset of hypertension, development of hypertension in patients aged <30 years.
2) Development of severe hypertension in patients >50 years.
3) Treatment-resistant or malignant hypertension.
4) Deterioration of renal function (eg, >30%) after the administration of an angiotensin-converting enzyme inhibitor (ACEI) or angiotensin receptor blocker (ARB).
5) Unexplained renal atrophy or a difference >1.5 cm in kidney size.
6) Unexplained progressive deterioration in kidney function.
7) Unexplained flash pulmonary edema.
8) Fibromuscular dysplasia of other vascular beds.
9) An epigastric or abdominal bruit.
2. Natural history: Over time significant renal artery stenosis, particularly when bilateral, may lead to the development of ischemic nephropathy and progressive chronic kidney disease (CKD). Fibromuscular dysplasia is also progressive and is associated with the development of new stenoses and arterial lesions (aneurysms and dissections).
DiagnosisTop
Diagnostic and management algorithm: Figure 1.
Most patients with atherosclerotic renal artery stenosis should be managed medically.Evidence 1Strong recommendation (benefits clearly outweigh downsides; right action for all or almost all patients). Moderate Quality of Evidence (moderate confidence that we know true effects of the intervention). Quality of Evidence lowered due to imprecision. Jenks S, Yeoh SE, Conway BR. Balloon angioplasty, with and without stenting, versus medical therapy for hypertensive patients with renal artery stenosis. Cochrane Database Syst Rev. 2014;(12):CD002944. doi: 10.1002/14651858.CD002944.pub2. Review. PubMed PMID: 25478936. Cooper CJ, Murphy TP, Cutlip DE, et al; CORAL Investigators. Stenting and medical therapy for atherosclerotic renal-artery stenosis. N Engl J Med. 2014 Jan 2;370(1):13-22. doi: 10.1056/NEJMoa1310753. PubMed PMID: 24245566; PubMed Central PMCID: PMC4815927. Investigation for renal artery stenosis should therefore be reserved for those with compelling indications to proceed with interventional approaches if renal artery stenosis is identified (see below).
1. Laboratory tests may reveal hypokalemia and albuminuria, although the latter is usually mild. An increase in serum creatinine levels and decrease in the GFR are frequently observed. Plasma renin activity (PRA) and aldosterone levels are often increased.
2. Imaging studies:
1) Duplex ultrasonography (renal Doppler ultrasonography may be the first line of imaging), computed tomography angiography (CTA), and magnetic resonance angiography (MRA) are reasonable initial noninvasive imaging modalities. Duplex ultrasonography may provide better functional versus anatomical information but is often limited due to the length of the procedure and lack of expertise. Limitations have been identified in the use of all noninvasive modalities, although many of the studies predate more recent advances in imaging quality. Ultimately, the choice of the initial test should be decided based on availability and local expertise, recognizing that most patients do not require imaging and can be managed medically. As a general rule, a positive test is more helpful than a negative one. CTA and MRA may miss lesions of fibromuscular dysplasia that are subsequently detected on digital substraction angiography (DSA).
2) Intra-arterial DSA is the most accurate method of imaging renal vasculature and is considered the gold standard. It is indicated in patients in whom the final diagnosis cannot be established using less invasive studies and there is an intent to proceed with an intervention if renal artery stenosis is confirmed.
3) The advantages of captopril renal scintigraphy are its noninvasive nature and the absence of nephrotoxic effects of radiopharmaceuticals (unlike in the case of radiographic contrast media). However, this test has fallen out of favor due to high false-positive and false-negative rates and limitations in those with preexisting CKD or bilateral renovascular disease.
3. Renal vein catheterization may be useful in determining indications for nephrectomy but is used infrequently. It allows for measuring PRA in the venous blood.
Diagnosis is based on the results of imaging studies.
Focus should be placed on primary and secondary hyperaldosteronism (hypokalemia) and renal parenchymal disease (renal failure).
TreatmentTop
1. Goals of treatment: Achievement of blood pressure targets (see Essential Hypertension). Treatment also includes modification of the factors predisposing to the development of atherosclerosis and treatment of comorbidities.
2. Treatment modalities (choice depends on the clinical manifestations and severity of renal artery stenosis [see below]):
1) Pharmacotherapy.
2) Revascularization: Percutaneous balloon angioplasty, percutaneous angioplasty with stenting, surgical correction of stenosis.
Pharmacotherapy may include:
1) Calcium channel blockers.
2) Beta-blockers: Their beneficial effect may be partially related to the inhibition of renin secretion.
3) Diuretics: Thiazide diuretics and/or a mineralocorticoid receptor antagonist (MRA) are often effective. Monitoring of kidney function and serum potassium levels is indicated when initiating.
4) ACEIs and ARBs are effective but may affect the function of the kidney supplied by the stenotic artery. These agents are contraindicated in patients with bilateral renal artery stenosis or renal artery stenosis of a solitary functioning kidney. Monitoring of kidney function is mandatory.
5) Antiplatelet therapy: Consider antiplatelet agents in patients with atherosclerotic renal artery stenosis treated with medical therapy provided there are no contraindications.Evidence 2Strong recommendation (benefits clearly outweigh downsides; right action for all or almost all patients). Low Quality of Evidence (low confidence that we know true effects of the intervention). Quality of Evidence lowered due to the observational nature of data and extrapolation of data from other cardiovascular territories. Ritchie J, Green D, Alderson HV, et al. Associations of antiplatelet therapy and beta blockade with patient outcomes in atherosclerotic renovascular disease. J Am Soc Hypertens. 2016 Feb;10(2):149-158.e3. doi: 10.1016/j.jash.2015.12.002. PubMed PMID: 26778771.
6) Lipid-lowering therapy (statins): Patients with atherosclerotic renal artery stenosis warrant treatment with statins provided there are no contraindications.Evidence 3Strong recommendation (benefits clearly outweigh downsides; right action for all or almost all patients). Moderate Quality of Evidence (moderate confidence that we know true effects of the intervention). Quality of Evidence lowered due to the lack of experimental evidence in this population but increased to moderate due to the use of lipid-lowering therapies in trials of optimal medical therapy and indirect evidence of benefit in other vascular territories.
1. Indications: Consider revascularization in patients with hemodynamically significant renal artery stenosis (stenosis of 80%-99% of the artery diameter, or of 60%-79% with a systolic pressure gradient across the stenosis ≥20 mm Hg; the latter is only measurable if the patient has already had intra-arterial angiography) and one of the following:
1) Coexisting uncontrolled hypertension despite maximal medical therapy.
2) Rapidly declining kidney function.
3) Acute pulmonary edema not explained by other etiology.
4) The etiology of the renal artery stenosis is fibromuscular dysplasia.
2. Techniques of revascularization:
1) Percutaneous renal angioplasty is the method of choice in most patients with renal artery stenosis caused by fibromuscular dysplasia. It is also effective in individuals with inflammatory stenosis (eg, Takayasu disease). The procedure may be performed only in patients in remission. Stenting is very rarely necessary and could impede the ability to proceed with surgical revascularization, when required.
2) Percutaneous renal angioplasty with stenting: In atherosclerotic renal artery stenosis, stenting is preferred over balloon angioplasty alone. In patients with stenosis caused by fibromuscular dysplasia, stenting is performed only in case of complications of angioplasty (arterial dissection).
3) Surgical renal revascularization is currently rarely performed and usually involves an aortorenal bypass graft. Occasionally surgical revascularization is required in lesions secondary to fibromuscular dysplasia, when angioplasty is not possible or ineffective.
3. Management of patients after percutaneous renal angioplasty:
1) Assess kidney function 24 hours after the procedure and then after 2 to 3 days. Carefully monitor blood pressure on the first day after the procedure because of the risk of hypotension.
2) Extrapolating from the interventional cardiology literature, the pattern of practice is to administer acetylsalicylic acid (ASA) 75 to 325 mg/d for lifetime; for the first 4 weeks after stent implantation, add clopidogrel 75 mg/d (in patients with implanted drug-eluting stents, consider extending clopidogrel therapy to 12 months).
3) To assess long-term effects of the procedure, continue to monitor blood pressure and renal function, initially more often and periodically thereafter.
Special ConsiderationsTop
1. Definition: A segmental, nonatherosclerotic, and noninflammatory disease of unknown etiology typically affecting the muscular layer of blood vessels, which causes stenosis of small and medium arteries. Fibromuscular dysplasia often presents with hypertension at a young age and is more common in women (a female to male ratio of ~4:1). The classical appearance on angiography is a “string-of-beads.”
2. Diagnosis: Indications for diagnostic workup of fibromuscular dysplasia: see Clinical Features, above. If first-degree relatives of a patient with fibromuscular dysplasia develop hypertension at a young age or have dissection of any artery, aneurysm, or cerebral bleeding, they should be evaluated for fibromuscular dysplasia.
Diagnostic tests:
1) Duplex ultrasonography (a potentially useful screening test with the same limitations as discussed above [see General Principles, above]).
2) MRA or CTA (the latter is preferred; Figure 2) to confirm the diagnosis or as a first-line study (in patients in whom duplex ultrasonography is presumed to be suboptimal [eg, patients with obesity]; these diagnostic methods are also used in the case of a very strong suspicion of fibromuscular dysplasia and/or serious clinical implications of the diagnosis [patients who are very young; patients with malignant hypertension or complications of hypertension, including complications affecting other vascular beds; patients with elevated serum creatinine levels]).
3) Intra-arterial DSA in patients with a strong suspicion of fibromuscular dysplasia when diagnosis remains equivocal after performing all the above studies or in patients with fibromuscular dysplasia confirmed by CTA or MRA in whom revascularization is indicated.
4) Differential diagnosis: Consider other rare diagnoses, including renal artery spasm; arteritis; and genetic diseases, such as type 1 neurofibromatosis, tuberous sclerosis, pseudoxanthoma elasticum, Ehlers-Danlos syndrome, Alagille syndrome, Williams syndrome, and Turner syndrome.
3. Treatment: Revascularization should be considered in most patients with hypertension and renal artery stenosis caused by fibromuscular dysplasia. Advancing age and longer duration of hypertension are negatively associated with improved outcomes after revascularization.Evidence 4Weak recommendation (benefits likely outweigh downsides, but the balance is close or uncertain; an alternative course of action may be better for some patients). Low Quality of Evidence (low confidence that we know true effects of the intervention). Quality of Evidence lowered due to the observational nature of studies. Trinquart L, Mounier-Vehier C, Sapoval M, Gagnon N, Plouin PF. Efficacy of revascularization for renal artery stenosis caused by fibromuscular dysplasia: a systematic review and meta-analysis. Hypertension. 2010 Sep;56(3):525-32. doi: 10.1161/HYPERTENSIONAHA.110.152918. Review. PubMed PMID: 20625080. Other scenarios in patients with fibromuscular dysplasia when revascularization should be considered include:
1) Recent-onset hypertension (this is the treatment of choice to normalize blood pressure).
2) Treatment-resistant hypertension or intolerance of antihypertensive drugs.
3) Renal failure or deteriorating renal function, particularly after the administration of an ACEI or ARB.
Preferred modalities of revascularization: see Revascularization, above.
In patients in whom revascularization is not performed:
1) Measure blood pressure monthly until target values are achieved.
2) Our pattern of practice is to measure serum creatinine (estimated glomerular filtration rate [eGFR]) levels and perform ultrasonography on a yearly basis to assess the kidney size.
In patients after revascularization:
1) Measure blood pressure and calculate the eGFR 1 month after the procedure.
2) Our pattern of practice is to perform renal imaging studies 6 months after the procedure, or earlier in case of elevated blood pressure or serum creatinine levels.
Tables and FiguresTop
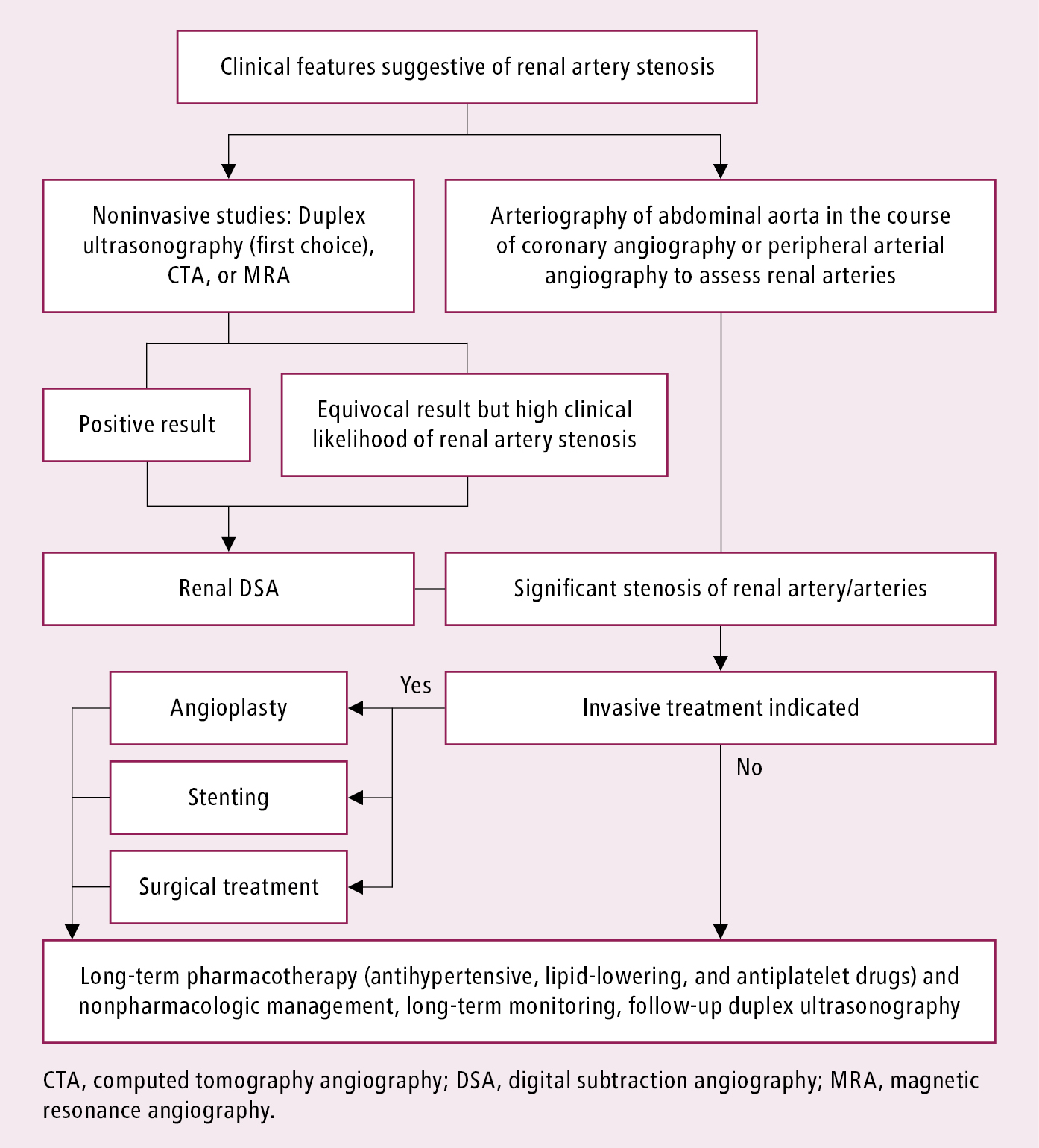
Figure 3.9-1. Management algorithm in patients with hypertension and renal artery stenosis. Adapted from Eur Heart J. 2018;39(9):763-816.
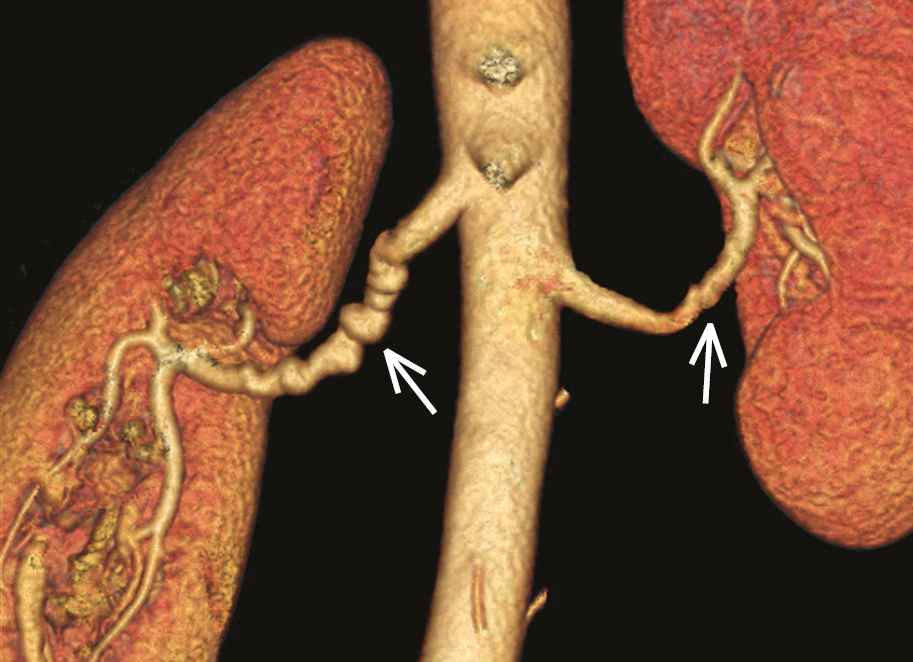
Figure 3.9-2. Computed tomography angiography (CTA) of a patient with fibromuscular dysplasia: the characteristic appearance of a “string of beads” in the right renal artery. Lesions on the left side are less advanced, without marked aneurysmal dilatations (arrows). Figure courtesy of Dr Aleksander Prejbisz.