Wildemberg LE, Fialho C, Gadelha MR. Prolactinomas. Presse Med. 2021 Oct 21;50(4):104080. doi: 10.1016/j.lpm.2021.104080. PMID: 34687915.
Zamanipoor Najafabadi AH, Zandbergen IM, de Vries F, et al. Surgery as a Viable Alternative First-Line Treatment for Prolactinoma Patients. A Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2020 Mar 1;105(3):e32-e41. doi: 10.1210/clinem/dgz144. PMID: 31665485; PMCID: PMC7112976.
Samperi I, Lithgow K, Karavitaki N. Hyperprolactinaemia. J Clin Med. 2019 Dec 13;8(12):2203. doi: 10.3390/jcm8122203. PMID: 31847209; PMCID: PMC6947286.
Wang AT, Mullan RJ, Lane MA, et al. Treatment of hyperprolactinemia: a systematic review and meta-analysis. Syst Rev. 2012 Jul 24;1:33. doi: 10.1186/2046-4053-1-33. PMID: 22828169; PMCID: PMC3483691.
Melmed S, Casanueva FF, Hoffman AR, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011 Feb;96(2):273-288. doi: 10.1210/jc.2010-1692. PMID: 21296991.
Klibanski A. Prolactinomas. N Engl J Med. 2010 Apr 1;362(13):1219-1226. doi: 10.1056/NEJMcp0912025. PMID: 20357284.
Casanueva FF, Molitch ME, Schlechte JA, et al. Guidelines of the Pituitary Society for the diagnosis and management of prolactinomas. Clin Endocrinol (Oxf). 2006 Aug;65(2):265-273. doi: 10.1111/j.1365-2265.2006.02562.x. PMID: 16886971.
Definition, Etiology, PathogenesisTop
Prolactin is a peptide hormone secreted by lactotroph cells of the anterior pituitary gland. Physiologically prolactin induces and maintains lactation. Its secretion is inhibited by dopamine released from the hypothalamus and stimulated by thyrotropin-releasing hormone (TRH), vasoactive intestinal peptide (VIP), epidermal growth factor, and estrogen (Figure 6.3-1).
Hyperprolactinemia is defined as a serum prolactin level above the upper limit of the reference range for nonpregnant, nonlactating individuals. Its prevalence in male patients is ~20 per 100,000, and in female patients, 90 per 100,000.
Causes of hyperprolactinemia:
1) Physiologic: Pregnancy, nursing/breastfeeding, breast stimulation, stress, exercise, sleep.
2) Pathologic:
a) Pituitary adenoma (prolactinoma or a mixed growth hormone and prolactin–secreting adenoma): Microadenoma <1 cm in size; macroadenoma >1 cm in size.
In prolactinomas the degree of prolactin elevation typically correlates with the size of the tumor. Prolactinomas are the most common functioning pituitary adenomas accounting for 40% to 50% of these tumors. Note that ~25% of somatotroph (growth hormone [GH]–secreting) tumors also cosecrete prolactin.
b) Medications: Dopamine antagonists (neuroleptics, domperidone, metoclopramide), tricyclic antidepressants, antiepileptics, verapamil, H2 blockers.
c) Primary hypothyroidism.
d) Renal failure.
e) Liver failure.
f) Polycystic ovary syndrome (PCOS).
g) Macroprolactinemia (big-big prolactin): A benign condition without clinical sequalae, caused by aggregation of prolactin monomers.
h) Stalk effect due to lesions in the hypothalamic area: Tumors, infiltrative disease, trauma, surgery in the region.
3) Idiopathic: No identifiable cause is found in 30% to 40% of cases.
Clinical Features and Natural HistoryTop
Clinical features may vary depending on the patient’s age, sex, etiology of hyperprolactinemia, and tumor size. Symptoms include hypogonadism (oligomenorrhea, amenorrhea, decreased libido, erectile dysfunction, infertility), galactorrhea, gynecomastia, and osteopenia. Bone loss occurs due to decreased levels of sex steroids resulting from hyperprolactinemia.
In the case of a large pituitary adenoma, symptoms of the mass effect may also manifest as headaches, visual field disturbances (eg, bitemporal hemianopsia if the optic chiasm is involved), diplopia, or hypopituitarism including symptoms of central adrenal insufficiency, hypothyroidism, and GH deficiency in addition to hypogonadism.
DiagnosisTop
If an elevated serum prolactin level above the upper limit of normal has been reported, testing should be repeated to confirm hyperprolactinemia.
The degree of prolactin elevation can help predict the etiology of pathologic hyperprolactinemia. Severe elevation >250 microg/L is typically due to a prolactinoma, but it may be caused by certain medications or primary hypothyroidism. Elevations >500 microg/L are invariably due to a prolactinoma. With hypothalamic stalk lesions, prolactin levels can vary from 25 to 200 microg/L. In other pathologic cases the prolactin level is usually <100 microg/L (Table 6.3-1).
In general the level of prolactin correlates with the size of the prolactinoma. With some laboratory assay techniques, very high antigen titers lead to assay artifacts, which may result in a major underestimation of prolactin levels (the so called hook effect), especially if the prolactinoma is large (eg, >3 cm) and the corresponding measured prolactin level is only 20 to 200 microg/L. The hook effect is currently becoming less of an issue in many laboratories.
Additional blood tests to establish the etiology (Figure 6.3-2):
1) TSH measurement to exclude primary hypothyroidism.
2) Creatinine/liver function tests to exclude renal/liver failure.
3) Beta-human chorionic gonadotropin (beta-hCG) measurement to exclude pregnancy.
4) Macroprolactin measurement (via polyethylene glycol [PEG] precipitation or gel filtration) to exclude macroprolactinemia.
5) Analysis for the hook effect, if suspected, by serial dilution prolactin testing (see above).
6) Anterior pituitary panel: Testosterone, estradiol, free thyroxine (FT4), luteinizing hormone (LH), follicle-stimulating hormone (FSH), adrenocorticotropic hormone (ACTH), cortisol, insulin-like growth factor 1 (IGF-1; for GH excess), as clinically indicated.
Imaging:
1) Magnetic resonance imaging (MRI) of the sella/pituitary to evaluate for prolactinoma and other sellar lesions.
2) Formal visual field testing if macroadenoma is large enough to compress the optic nerves or chiasm.
TreatmentTop
Treatment of hyperprolactinemia is indicated if the disease is symptomatic (eg, menstrual irregularities, amenorrhea, hypogonadism, galactorrhea), associated with structural compressive neurologic symptoms (due to compression of the optic chiasm and extraocular nerves by the tumor), or in the treatment of infertility.
It is important to treat the underlying cause of hyperprolactinemia if possible.
In the case of a prolactinoma, especially a macroprolactinoma, specific treatment (usually a dopamine agonist) is often necessary due to the risk of compressive neurologic symptoms and hypopituitarism. It is important to treat the underlying cause if possible.
For microprolactinomas specific treatment is indicated if fertility is desired or if galactorrhea is bothersome to the patient. In hyperprolactinemia causing hypogonadism, testosterone treatment can be considered in men who do not tolerate or respond to dopamine agonists, if they do not desire fertility. In women who do not want to conceive, estradiol with a progestin in the form of an oral contraceptive or low-dose hormonal therapy can be considered as a sole therapy for hypogonadism in those who do not tolerate or respond to dopamine agonists. Estradiol should not be used as the sole treatment for macroprolactinomas.
Oral contraception or combination of estradiol and progesterone are also options for women who have hypogonadism resulting from hyperprolactinemia due to other causes, including antipsychotic agent use.
For drug-induced hyperprolactinemia, no treatment is necessary if the condition is asymptomatic. If it is symptomatic, consider discontinuing the drug and trialing an alternative provided it is clinically safe. If the patient uses an antipsychotic, an alternative such as quetiapine, olanzapine, ziprasidone, or aripiprazole, which do not raise serum prolactin levels, can be considered in discussion with the patient’s mental-health professional. If the drug cannot be discontinued, treatment of hypogonadism with estrogen or testosterone should be considered.
Hyperprolactinemia due to macroprolactinemia (big-big prolactin) does not require treatment, and hyperprolactinemia due to underlying primary hypothyroidism should be treated with thyroxine.
First-line treatment for prolactinomas is usually medical therapy (not surgery) with dopamine agonists, which have a 50% to 90% success rate at decreasing serum prolactin levels and the size of the prolactinoma. Dopamine agonists (cabergoline, bromocriptine, and quinagolide) inhibit the secretion of prolactin from the anterior pituitary. Their adverse effects include nausea, headache, dizziness, abdominal pain, orthostasis, fatigue, and syncope. Sometimes they can lead to onset or worsening of impulse control disorders, valvular heart disease (cabergoline), or cerebrospinal fluid rhinorrhea/leak if used in the treatment of large adenomas or in higher doses. Cabergoline is generally preferred over bromocriptine as first-line therapy owing to a better adverse effect profile, convenient dosing regimen, and greater efficacy.
Dosing: Titrate the dose depending on symptoms, prolactin levels, and tumor size change:
1. Cabergoline: Start at 0.25 to 0.5 mg/wk administered in 1 or 2 divided weekly doses; you may increase the dose by 0.25 to 0.5 mg/wk no sooner than every 4 weeks, up to a maximum dose of 3 mg/wk.
2. Bromocriptine: Start at 1.25 to 2.5 mg nightly; you may increase the dose by 2.5 mg/d every 2 to 7 days, as tolerated, up to 15 mg/d administered in 1 or 2 divided doses per day.
3. Quinagolide: Start at 25 microg once daily for 3 days followed by 50 microg once daily and by further stepwise titration by 75 to 150 microg/d at intervals ≥4 weeks, up to a maximum of 900 microg/d.
Monitoring and follow-up: Serum prolactin should be measured 1 month after therapy initiation. The dopamine agonist should be continued at the initial dose if the prolactin level has normalized, symptoms have improved, and tumor size has decreased. If the prolactin level has decreased but not yet normalized and symptoms persist, the dose should be increased, provided that no intolerable adverse effects are reported, to achieve symptom resolution and reduction in tumor size. For macroprolactinomas (>1 cm) MRI should be repeated within 4 to 12 months to reassess tumor size. Visual field testing should be repeated within 1 to 2 months if its result was initially abnormal. For patients on >2 mg/wk of cabergoline, clinical monitoring, echocardiography, or both should be performed periodically to screen for valvular heart disease.
If the patient’s response is inadequate or intolerable adverse effects of a dopamine agonist occur, an alternative dopamine agonist can be trialed. If therapy with >1 dopamine agonist is unsuccessful or not tolerated, transsphenoidal surgery (for prolactinomas) or sex hormone replacement therapy for hypogonadism can be considered.
Withdrawal or tapering of therapy after 2 years may be pursued in patients with stable prolactinomas with close clinical, biochemical, and radiologic monitoring if prolactin levels are low to normal and the tumor is radiographically responsive (no or a minimal tumor on MRI). Tapering of the dopamine agonist should be slow and gradual.
Surgical resection of a prolactinoma by an experienced pituitary surgeon is considered in cases refractory to medical therapy (failure to achieve normal prolactin and a 50% decrease in tumor size), patients intolerant to medical therapy, pregnant patients with macroprolactinomas that may affect vision or are nonresponsive to medication, or based on patient preferences. Surgical cure occurs in 75% of microadenoma and in 25% of macroadenoma cases. Surgical complications include hypopituitarism, vasopressin disorders (previously known as diabetes insipidus), and risks of infection and bleeding.
Of note, some authors suggest surgery as a primary modality of treatment.Evidence 1Low Quality of Evidence (moderate confidence that we know true effects of the intervention). Quality of Evidence lowered due to the risk of bias, especially in the setting of a microprolactinoma or an intrasellar macroprolactinoma with access to a high-volume pituitary surgeon. Zamanipoor Najafabadi AH, Zandbergen IM, de Vries F, et al. Surgery as a Viable Alternative First-Line Treatment for Prolactinoma Patients. A Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2020 Mar 1;105(3):e32–41. doi: 10.1210/clinem/dgz144. PMID: 31665485; PMCID: PMC7112976. De Sousa, S. M. C. Dopamine agonist therapy for prolactinomas: do we need to rethink the place of surgery in prolactinoma management? Endocrine Oncology. 2022;2(1), R31-R50. doi.org/10.1530/EO-21-0038.
Radiation therapy has a ~25% success rate, but it may be complicated by hypopituitarism, optic nerve damage, and cognitive dysfunction. Radiation therapy is primarily used to prevent regrowth of a residual tumor in a patient with a history of transsphenoidal debulking of a very large macroadenoma resistant to medical therapy, or for an aggressive/malignant prolactinoma resistant to medical therapy, which cannot be treated with primary surgery.
Treatment of Refractory Malignant Prolactinomas
A prolactinoma may be considered refractory when dopamine agonists at high doses do not allow to achieve normal prolactin levels or to reduce tumor size by 50%, are not tolerated, and surgical resection/radiotherapy is unsuccessful or is followed by rapid disease recurrence and progression. As these cases are often characterized by rapid growth and may metastasize, they are termed malignant prolactinomas. The oral cytotoxic chemotherapy agent temozolomide may be used in the treatment of refractory malignant prolactinomas.
PrognosisTop
In patients with prolactinomas dopamine agonists have a 50% to 90% success rate of normalizing serum prolactin levels, reversing symptoms, and restoring gonadal function and fertility. Drug discontinuation may be considered after stabilization with 2 years of therapy, but recurrence of hyperprolactinemia after withdrawal is common. Long-term medical therapy is generally required in these patients.
Tables and FiguresTop
|
Cause of prolactin elevation |
Typical range in serum |
|
Idiopathic Pituitary stalk interruption Primary hypothyroidism Renal failure Liver failure Medications |
20-100 ng/mL |
|
Prolactinoma Medications Pituitary stalk interruption Primary hypothyroidism |
100-250 ng/mL |
|
Prolactinoma Medications Primary hypothyroidism |
250-500 ng/mL |
|
Prolactinoma |
>500 ng/mL |
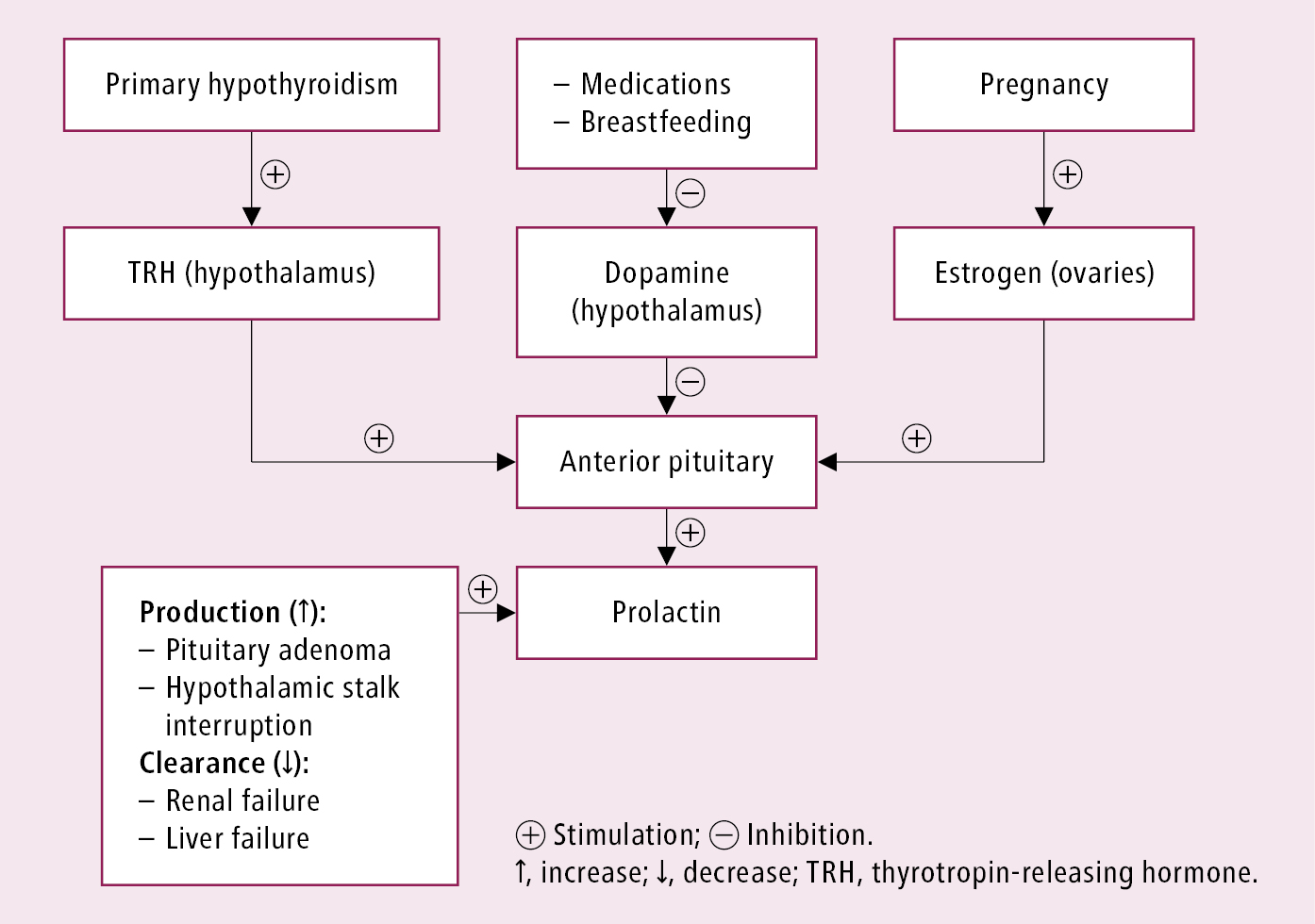
Figure 6.3-1. Stimulators and inhibitors of prolactin secretion. Adapted from CMAJ. 2003 Sep 3;169(6):575-581.
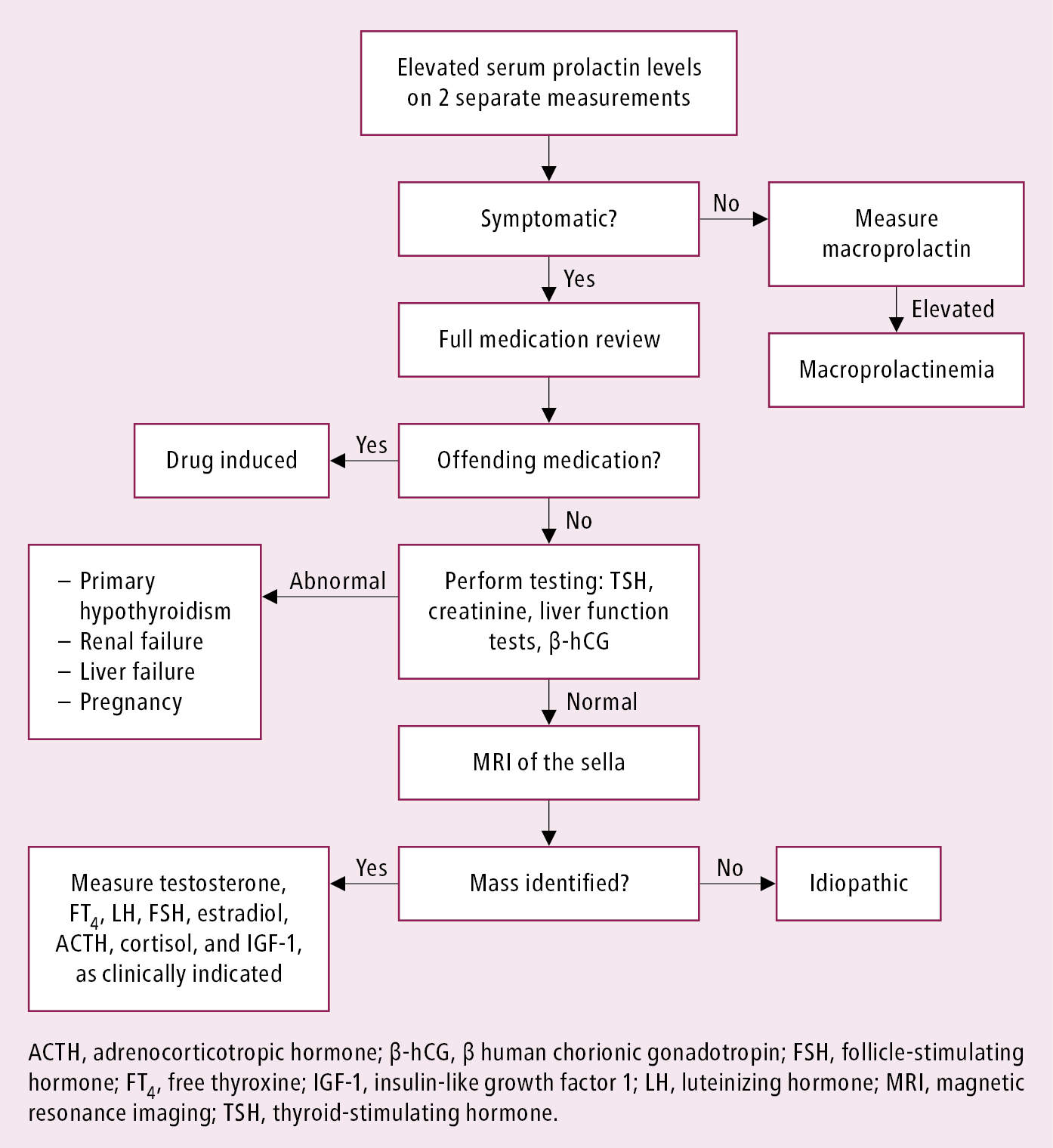
Figure 6.3-2. Diagnostic algorithm for hyperprolactinemia.