ELSO Guidelines for Cardiopulmonary Extracorporeal Life Support Extracorporeal Life Support Organization, Version 1.4. August 2017. Ann Arbor, MI, USA.
Rihal, CS, Naidu SS, Givertz MM, et al. 2015 SCAI/ACC/HFSA/STS Clinical Expert Consensus Statement on the Use of Percutaneous Mechanical Circulatory Support Devices in Cardiovascular Care. JACC. 2015 May 65(19):e7-26. doi: http://dx.doi.org/10.1016/j.jacc.2015.03.036. PMID: 25861963.
Extracorporeal membrane oxygenation (ECMO) provides cardiopulmonary support for patients with acute severe heart or respiratory failure despite conventional therapy. The ECMO system consists of a membrane oxygenator to perform gas diffusion, a roller or centrifugal pump to achieve blood flow through the system, heat exchanger, pressure and temperature sensors, and a drainage system. Vascular access is provided by cannulation of large peripheral vessels (the femoral or internal jugular vein and the femoral, axillary, or subclavian artery). Cannulation can be performed using a percutaneous or surgical technique.
Types of ECMO systems (Figure 21.10-1):
1) Venovenous ECMO (VV-ECMO) allows for collection of deoxygenated blood from the venous system and transfer of oxygenated blood to the right atrium. Double-lumen cannulas are most commonly used to collect and transfuse blood through a single vascular access site (eg, the internal jugular vein).
2) Venoarterial ECMO (VA-ECMO) allows for collection of deoxygenated blood from the venous system and transfer of oxygenated blood directly into the arterial system. This can be achieved through central or peripheral cannulation via a large peripheral artery (eg, femoral, axillary, or subclavian).
VV-ECMO enables extracorporeal oxygenation of the blood, while VA-ECMO ensures simultaneous extracorporeal oxygenation of the blood and adequate blood flow through the arterial system. In patients with biventricular failure, VA-ECMO is the mechanical circulatory support of choice for those with cardiogenic shock and impaired oxygenation because it provides full cardiopulmonary support. With peripheral VA-ECMO, due to the retrograde flow support, blood travels in the direction opposite to normal (ie, from the femoral artery back toward the thoracic aorta), which may increase left ventricular afterload.
It is important to note that as VV-ECMO relies on the patient’s native cardiac function for circulation, it does not provide any hemodynamic support.
The scope of cardiac support depends on the predetermined flow generated by VA-ECMO and can be individually adjusted to the patient’s needs. Anticoagulation is required during therapy, usually with IV unfractionated heparin, controlled with activated clotting time (ACT), activated partial thromboplastin time (aPTT) (usually measured ≥3 times a day), or both.
IndicationsTop
Indications for VA-ECMO:
1) Cardiogenic shock.
2) Acute heart failure (HF) (INTERMACS 1 and 2; see Mechanical Circulatory Support (MCS)).
3) Low cardiac output syndrome following cardiac surgery.
4) Extracorporeal cardiopulmonary resuscitation (ECPR).
VA-ECMO can be used as a bridge to decision (BTD), bridge to bridge (BTB), bridge to transplant (BTT), as well as bridge to recovery (BTR).
As stated in the 2021 European Resuscitation Council (ERC), ECMO for ECPR may be considered as rescue therapy in selected patients with cardiac arrest when conventional cardiopulmonary resuscitation (CPR) is failing. Commonly used criteria include witnessed cardiac arrest with bystander CPR, short time to establish ECPR (<60 min from CPR onset), young age with no major comorbidities, and a known or suspected treatable cause of cardiac arrest. ECPR should be considered particularly in hypothermic cardiac arrest as well as hypothermia and risk factors for imminent cardiac arrest (ie, core temperature <30 degrees Celsius, ventricular arrhythmia, systolic blood pressure <90 mm Hg). However, prognostic evaluation of successful rewarming should be assessed whenever possible based on the Hypothermia Outcome Prediction after Extracorporeal Life Support (HOPE) (www.hypothermiascore.org).
According to the 2017 Extracorporeal Life Support Organization (ELSO) guidelines for respiratory failure in adults, VV-ECMO should be considered in severe isolated respiratory failure without HF and with a high risk of death (see Acute Respiratory Failure). During the coronavirus disease 2019 (COVID-19) pandemic, ECMO applied in the treatment of COVID-19–related respiratory failure became the dominant use of this approach. At that time VA-ECMO continued to be used as both respiratory and cardiac output support or replacement.
ContraindicationsTop
ECMO is used in emergent cases as rescue therapy. Therefore, contraindications include irreversibility of the underlying disease, absolute contraindications to anticoagulation, and severe immunosuppression. In the case of VV-ECMO used for isolated respiratory support, contraindications in our own setting include intensive mechanical ventilation for ≥7 days (high fraction of inspired oxygen [FiO2] and high airway pressures, eg, plateau pressure >30 cm H2O). In addition, the risk of severe complications increases significantly with age.
ComplicationsTop
1. Injury to the vessel and bleeding from cannulation sites.
2. Neurologic complications (seizure, hemorrhagic or ischemic stroke).
3. Complications of anticoagulation (bleeding, heparin-induced thrombocytopenia), and transfusion reactions.
4. Blood clotting in the ECMO circuit resulting in consumption of clotting factors and platelets.
5. Mechanical damage to blood components (hemolysis, anemia, thrombocytopenia).
6. Renal failure requiring renal replacement therapy.
7. Bloodstream infections, sepsis.
8. Systemic inflammatory response syndrome (SIRS) due to the contact of blood with the surface of the ECMO circuit (this may cause acute respiratory distress syndrome [ARDS]).
9. Altered pharmacokinetics of drugs (increased elimination due to binding in the ECMO circuit).
10. Other VA-ECMO complications: Limb ischemia (due to insertion of an arterial cannula; can be managed with insertion of an additional reperfusion cannula), hypoxia of the upper body (Harlequin syndrome may occur when a cannula that provides oxygenated blood is inserted through the femoral artery in a patient with respiratory failure; can be managed with insertion of an additional cannula to provide oxygenated blood from the ECMO circuit through the internal jugular vein [veno-arterial-venous ECMO, or VAV-ECMO]), worsening of left ventricular failure and pulmonary edema (eg, with too high blood flow through the ECMO circuit), pulmonary hypoxia.
FigureSTop
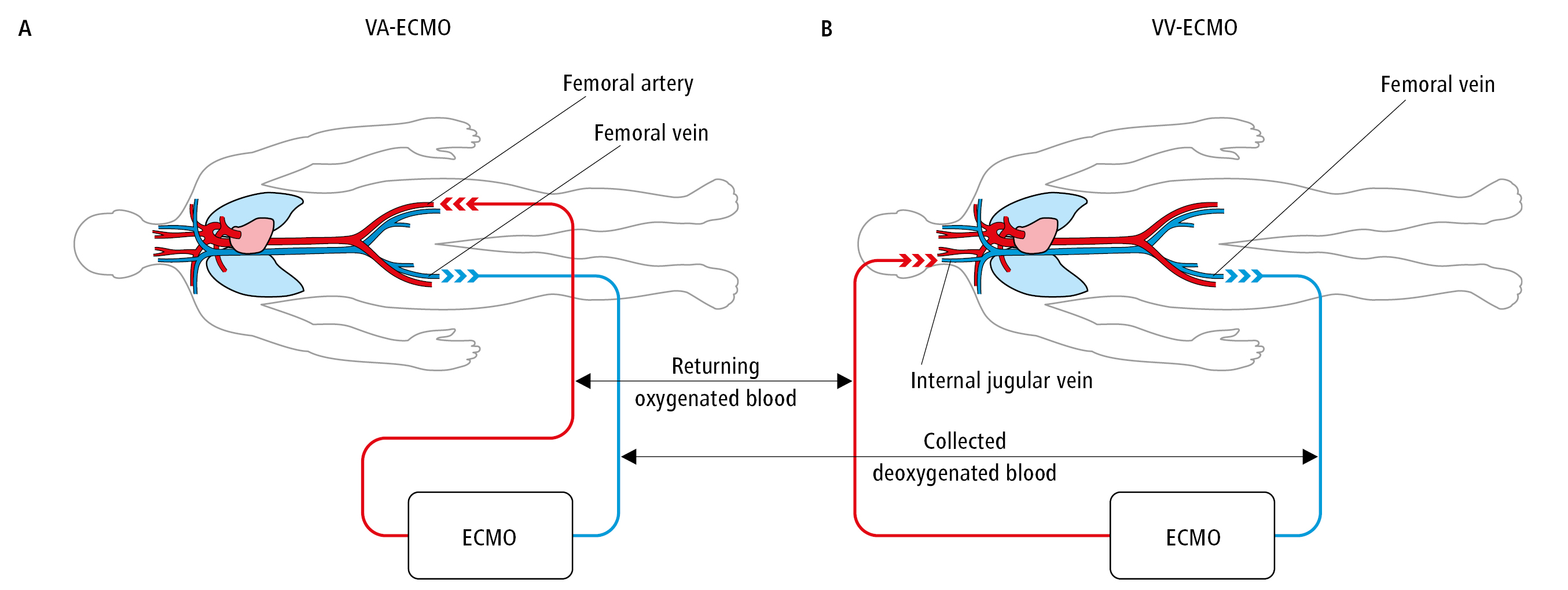
Figure 21.10-1. Extracorporeal membrane oxygenation (ECMO). A, venoarterial system (VA-ECMO). B, venovenous system (VV-ECMO).