References
Lee CH, Steiner T, Petrof EO, et al. Frozen vs Fresh Fecal Microbiota Transplantation and Clinical Resolution of Diarrhea in Patients With Recurrent Clostridium difficile Infection: A Randomized Clinical Trial. JAMA. 2016 Jan 12;315(2):142-9. doi: 10.1001/jama.2015.18098. PubMed PMID: 26757463.Moayyedi P. Fecal transplantation: any real hope for inflammatory bowel disease? Curr Opin Gastroenterol. 2016 Jul;32(4):282-6. doi: 10.1097/MOG.0000000000000285. PubMed PMID: 27152872.
Roman Jaeschke: Are we at the stage where we think about manipulating our own microbiome in any way?
Paul Moayyedi: We certainly are doing that. Fecal transplant is the new kid on the block here. You give a person who has a disease the fecally contaminated water from someone who has been screened and is healthy in all aspects that you can tell.
RJ: Sorry to ask you the question, but how do we give it?
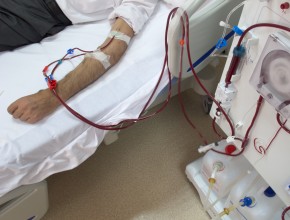
PM: Good question, because it varies depending on the person. We do this at McMaster University, particularly Dr. Christine Lee, who is an infectious diseases person at McMaster. Basically, it is given by retention enema – 50 mL of fecally contaminated water through the rectum. The patient holds it for 20 minutes and then can go to the bathroom. That is how we use it. Some others give it colonoscopically: they basically give you a colonoscope and squirt it through the channel of the colonoscope. Others still give it via a nasojejunal tube: you pass a tube up your nose into the jejunum, so it bypasses the stomach and is given that way. It is not so pleasant and so is coming more out of favor. Pills have also been developed, but again these are still in the development phase.
RJ: Do we know something about efficacy?
PM: Yes. In Clostridium difficile we do know something about the efficacy. This is where it was first used; in fact, it was used as early as the 1950s to treat antibiotic resistance of C difficile. Indeed, as a resident in the 1990s I used this to treat a person who was very sick and it was miraculous. Others have noticed this as well and now we have large case series. I did a review of the literature for the Canadian Association of Gastroenterology and we had about 20 case series showing that it worked in about 90% of cases that were resistant to antibiotics. We also have one randomized trial, which also shows that it works better than vancomycin in these antibiotic-resistant individuals.
RJ: Is one fecal transplant working as well as another fecal transplant is working or is it donor-specific somehow?
PM: For C difficile there seems be a very wide therapeutic window. Any donor will do. Any route will do. You can give it rectally, you can give it via this nasogastric tube, but either way it seems to work. Just one donation is usually enough, although with C difficile you may need to give another after a week or so if nothing is working. It seems to be remarkably and easily effective. This is why even in an evidence-based world we do not need masses of randomized trials here because the effect is so marked.
RJ: So you get a donor card as well?
PM: The big thing about the donor is they have to be healthy. We choose people without any psychiatric comorbidities, they cannot be obese or have any other illnesses, and their stool is heavily screened for any agent that could cause harm in the future. Of course we do not know everything, so the danger of this is that we may be transmitting things that we do not know yet, very much as we did with hepatitis C and blood transfusions in time gone by. The advantage to this as opposed to the hepatitis C analogy is that we are not giving it directly into the circulation. As I said before, the immune system is focused on the gut, so even if we do give something that could potentially be harmful it may well be that host can maintain that. I heard that [in the case of] the first fecal transplant that one of my very much older colleagues did in the 1950s, the donor actually had typhoid in the end. The person was fine and did not develop typhoid. It is probably a little more robust than passing agents through blood but nevertheless it is a risk we have yet to determine.
RJ: What an interesting world we live in. We were talking about microbiome: you told us what it is and about one example of manipulating the microbiome. I have heard something today on the topic of inflammatory bowel disease and changing the microbiome. Could you expand here?
PM: We have a lot of agents already that treat inflammatory bowel disease, Crohn disease, and ulcerative colitis, but none of them are perfect. Most of them require you to keep taking the product to get into long-term remission, and even then you get relapses, so our treatment is yet to be perfect. With this understanding of the microbiome has come the concept that rather than focus on drugs that suppress the immune system, which is what we have heard today, you look at what is driving that immune system in the first place. The obvious thing is your microbiome, because it is just sitting right there and the immune system is sitting there trying to protect you from it, so if that balance goes wrong it may well be that is the cause of ulcerative colitis and Crohn disease.
We have looked at fecal transplants in ulcerative colitis in a randomized trial, which we published recently, and we found that fecal transplants were more effective than placebo at inducing remission at week 7: roughly 24% of people were in remission in the fecal transplant arm, 5% in the placebo.
RJ: So 1 in 5.
PM: Yes. This is not a miracle by any means but we are just starting, so we will now look at, for example, donor-specificity that you mentioned before. It does seem that in inflammatory bowel disease there may be donor-specificity. There was a particular donor that worked well and one which did not work at all. It may well be that in ulcerative colitis you need specific donors. We are certainly looking at that in the next trial to see if we can make this work better.
It is important to note that if you look at the end points we use, the current therapies for pure remissions at week 7 only work in about 24% of cases as well. This is early data – only 75 patients – but it is promising that this therapy may become in the future a treatment for ulcerative colitis.
RJ: For such persistent, chronic, and sometimes debilitating disease, a 20% reduction in absolute terms is actually quite amazing.
PM: Yes.
This interview was recorded at the 2nd McMaster International Review Course in Internal Medicine in Kraków, Poland. To learn more, visit www.mircim.eu.
 English
English
 Español
Español
 українська
українська