Atraumatic vs conventional lumbar puncture needles
Atraumatic and conventional needles.
Performing lumbar puncture (LP) using the so-called atraumatic (cone-shaped) needles rather than conventional ones results in fewer headaches and decreased rates of other adverse consequences.
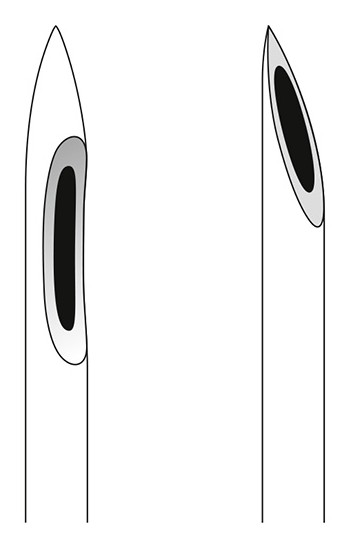
Conventional needles have a sharp slanted tip with a distal opening, whereas atraumatic needles have a pencil point tip and a side port opening.
This meta-analysis comprised 110 clinical studies involving over 31,400 patients undergoing LP. Most procedures were done for spinal anesthesia, and the rest was performed mostly for diagnostic indications and myelography. Headache occurred in 4.2% of patients having LP done using an atraumatic needle and 11% of people whose LP was done using a conventional needle (relative risk [RR], 0.40; 95% CI, 0.34-0.47). The need for intravenous fluid or controlled analgesia was decreased from 4.5% to 2.2%, and the need for epidural blood patch was decreased from 3.0% to 1.1%. The type of needle has not influenced the success of LP on first attempt, failure rate, and occurrence of backache or traumatic tap.
Unpublished information obtained by the authors suggests that awareness of the benefit of atraumatic needles is higher among anesthetists than among internists or neurologists.
Bright light therapy in bipolar depression

Carex Day-Light Classic. www.carex.com.
Bright light therapy at midday may have major beneficial effects in some patients with depression in the setting of bipolar mood disorder.
Forty-six patients with a current moderate to severe episode of depression and diagnosed bipolar disorder were randomized to bright light or placebo dim red light. Therapy was delivered between noon and 14:30, lasting from 15 minutes initially to 60 minutes daily (duration was increased weekly by 15 minutes). It used a 4000-K broad-spectrum white fluorescent light (Carex Day-Light Classic) with an intensity of 7000 lux and a 50-lux red light as a control.
At 4 to 6 weeks the remission rate of depression was 68.2% in the bright light therapy group versus 22.2% in the control group. There were no episodes of hypomania or mood polarity switch. Such results indicate that 1 in 2 patients would benefit from this type of therapy.
Complete control of severe hemophilia A after a single infusion
van den Berg HM. A Cure for Hemophilia within Reach. N Engl J Med. 2017 Dec 28;377(26):2592-2593. doi: 10.1056/NEJMe1713888. Epub 2017 Dec 9. PubMed PMID: 29224412.
Hemophilia A may be completely controlled for at least a year after a single drug infusion.
In this dose-finding study 9 patients with severe hemophilia A received a single infusion of a medication encoding factor VIII (for those interested: a codon-optimized adeno-associated virus serotype 5 [AAV5] vector encoding a B-domain-deleted human factor VIII [AAV5-hFVIII-SQ]). One patient received a low dose, and one patient received an intermediate dose of the medication. A high dose was delivered to 7 patients; in all participants in this group, the factor VIII activity level increased from <1 international units/dL to >5 international units/dL by week 9, and in 6 out of 7 the level was normalized (>50 international units/dL) and maintained at 1 year.
Prior to the study, all patients had been receiving prophylactic factor VIII therapy yet had a median annualized bleeding rate of 16 events; this declined to 1 event per year after the infusion. The use of factor VIII was stopped in all 7 participants by week 22.
 English
English
 Español
Español
 українська
українська