Lembo A, Sultan S, Chang L, Heidelbaugh JJ, Smalley W, Verne GN. AGA Clinical Practice Guideline on the Pharmacological Management of Irritable Bowel Syndrome With Diarrhea. Gastroenterology. 2022 Jul;163(1):137-151. doi: 10.1053/j.gastro.2022.04.017. PMID: 35738725.
Arasaradnam RP, Brown S, Forbes A, et al. Guidelines for the investigation of chronic diarrhoea in adults: British Society of Gastroenterology, 3rd edition. Gut. 2018 Aug;67(8):1380-1399. doi: 10.1136/gutjnl-2017-315909. Epub 2018 Apr 13. PMID: 29653941; PMCID: PMC6204957.
Shane AL, Mody RK, Crump JA, et al. 2017 Infectious Diseases Society of America Clinical Practice Guidelines for the Diagnosis and Management of Infectious Diarrhea. Clin Infect Dis. 2017 Nov 29;65(12):1963-1973. doi: 10.1093/cid/cix959. PMID: 29194529; PMCID: PMC5848254.
Riddle MS, DuPont HL, Connor BA. ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Acute Diarrheal Infections in Adults. Am J Gastroenterol. 2016 May;111(5):602-22. doi: 10.1038/ajg.2016.126. Epub 2016 Apr 12. PMID: 27068718.
Definition and PathogenesisTop
Diarrhea is arbitrarily defined as passing excessively loose (liquid or semiliquid) stools with increased frequency (≥3 per day), increased volume (200-250 g per day), or both.
1. Mechanisms:
1) Impaired absorption from the small or large intestine caused by:
a) Loss of mucosal surface or damage to the epithelial absorption mechanism.
b) Presence of nonabsorbable osmotically active substances in the intestinal lumen (osmotic diarrhea), which cause movement of fluids into the intestinal lumen driven by the osmotic gradient.
c) Accelerated gastrointestinal (GI) transit (increased motility).
2) Increased secretion of water and electrolytes into the lumen of the small or large intestine (secretory diarrhea) due to activation of transport mechanisms in the intestinal epithelium or of the intestinal nerves by:
a) Enterotoxins.
b) Inflammatory mediators (adenosine, histamine, serotonin, hydrogen peroxide, platelet-activating factor [PAF], leukotrienes, prostaglandins, cytokines). Inflammatory diarrhea is often accompanied by malabsorption resulting from epithelial injury and decreased mucosal surface.
c) Gut hormones.
2. Classification and causes: Diarrhea can be classified based on pathophysiology, anatomic location, histopathology, duration of symptoms, etiology (organic or functional), volume (large or small), and whether it is bloody or nonbloody. On the basis of its duration, diarrhea can be classified as acute (duration ≤14 days), persistent (duration >14 but <30 days), or chronic (duration >30 days). Note that these definitions are arbitrary and vary among professional societies and practice guidelines. For example, acute diarrhea may be defined as lasting from <7 to <14 days, whereas chronic diarrhea may be defined as lasting >28 days or >30 days. Persistent diarrhea is defined as lasting between 14 and 30 days. Some organizations have also defined prolonged diarrhea (7-13 days).
The Bristol stool scale (Figure 1.9-1) can be used to assess stools based on their appearance.
1) Acute diarrhea:
a) GI tract infections or ingestion of bacterial toxins (>90% of cases of acute diarrhea).
b) Adverse drug reactions: It can be challenging to identify the agent causing drug-induced diarrhea, particularly in patients taking multiple agents. The clinical setting and timing of the onset of symptoms relative to when the medications were started can be helpful. The most frequent cause of noninfectious diarrhea are broad-spectrum antibiotics, antiarrhythmics (beta-blockers, diltiazem), antihypertensive agents (eg, angiotensin-converting enzyme inhibitors [ACEIs], diuretics), nonsteroidal anti-inflammatory drugs (NSAIDs), theophylline, antidepressants (selective serotonin reuptake inhibitors), cytotoxic agents, H2 blockers, antacids (magnesium hydroxide), metformin, thyroid hormones, and laxative abuse (diarrhea usually resolves within 24-48 hours of discontinuing the laxative).
c) Toxins: Mushroom poisoning (eg, poisoning with alpha-amanitin from the death cap), insecticides (organic phosphates), ethanol, and arsenic.
d) Food hypersensitivity.
e) Ischemic colitis and acute colonic diverticulitis.
2) Chronic diarrhea (>4 weeks) is rarely caused by GI infections (with the exception of immunocompromised patients). In the vast majority of cases the causes are inflammatory bowel disease (IBD), colon cancer, or irritable bowel syndrome (IBS). Classification based on pathophysiology can be helpful in narrowing down differential diagnosis and is well described by the following mechanisms: secretory diarrhea, osmotic diarrhea, steatorrhea, inflammatory diarrhea, accelerated GI transit.
a) Secretory diarrhea: Common causes of secretory diarrhea include toxins (chronic alcohol use disorder, arsenic), bile acids (impaired bile acid absorption in the ileum due to, eg, bacterial overgrowth syndrome; terminal ileitis; resection of the terminal ileus), short-chain fatty acids, microscopic colitis, collagenous colitis, functioning endocrine tumors (carcinoid, VIPoma, gastrinoma, villous adenoma of the colon, medullary thyroid carcinoma, mastocytosis). Drugs: The most frequent causes are contact laxatives (bisacodyl, anthranoids, aloe vera) and other laxatives (as in acute diarrhea), antiarrhythmics (quinidine, digoxin), auranofin, caffeine, calcitonin, carbamazepine, and chemotherapeutic agents. Other potential causes include cimetidine, misoprostol, metformin, NSAIDs, simvastatin, theophylline, levodopa/benserazide, and cholinesterase inhibitors.
b) Osmotic diarrhea: Certain food and confectionery products that contain fructose, sorbitol, mannitol, or xylitol; lactase deficiency (lactose intolerance) and other types of disaccharidase deficiency (primary [congenital; eg, adult-type hypolactasia] or secondary [eg, to intestinal infection or inflammation]); short bowel syndrome; intestinal fistula and enteral feed. Drugs: Osmotic laxatives (magnesium sulfate, polyethylene glycol, lactulose); antacids (magnesium hydroxide); orlistat, acarbose, cholestyramine, ampicillin, clindamycin, neomycin, phosphates, prebiotics, quinidine, propranolol, hydralazine, ACEIs, procainamide, biguanides, methyldopa.
c) Steatorrhea: Maldigestion (exocrine pancreatic insufficiency [chronic pancreatitis, pancreatic cancer, cystic fibrosis], bacterial overgrowth syndrome, cholestatic liver disease), malabsorption (celiac disease, giardiasis, Whipple disease, bowel ischemia, abetalipoproteinemia, intestinal lymphangiectasia, and other causes of protein-losing enteropathy).
d) Inflammatory diarrhea: IBD (Crohn disease, ulcerative colitis); microscopic, ischemic, or radiation-induced colitis (eg, after radiotherapy to the abdominal region); food hypersensitivity; primary and secondary immunodeficiency; intestinal tumors (eg, colon cancer); intestinal protozoal infections (Giardia intestinalis, Entamoeba histolytica, Cryptosporidium parvum, Isospora spp, Cyclospora spp) and helminthiasis.
e) Drug-induced diarrhea: Antibiotics (clindamycin, ampicillin, amoxicillin, cephalosporins), auranofin, carbamazepine, etanercept, flutamide, cytotoxic agents, cyclosporine (INN ciclosporin), NSAIDs, statins, mercaptopurine, mycophenolate mofetil, olmesartan, oral contraceptives, penicillamine, rituximab, selective serotonin reuptake inhibitors (SSRIs), sodium phosphate, ticlopidine, tyrosine kinase inhibitors, H2 blockers, proton pump inhibitors (PPIs), gold salts, and laxatives.
f) Accelerated GI transit (ie, increased motility): Thyrotoxicosis, carcinoid, and diabetes mellitus. Drugs: Prokinetic agents (metoclopramide, cisapride), acetylcholinesterase inhibitors (tacrine), cholinergic agents (bethanechol), irinotecan, macrolides (erythromycin), ticlopidine, thyroid hormones, colchicine.
g) Functional diarrhea: IBS—a disorder of the gut-brain axis fulfilling the Rome IV criteria, with the absence of alarm features.
Selected aspects of diarrhea in patients with cancer: In patients receiving palliative care, diarrhea is most frequently due to:
1) Overtreatment with laxatives (often in patients with inadequately treated constipation).
2) GI infections.
3) Cytotoxic agents (most frequently fluorouracil, irinotecan, mitomycin) and radiotherapy to the abdomen or pelvis.
4) Enteral nutrition.
5) Insufficient pancreatic exocrine secretion (steatorrhea) in patients with pancreatic head tumors.
6) Bile acid and carbohydrate malabsorption (disaccharidase deficiency) after ileal resection or cholecystectomy. In such patients diarrhea is caused by excessive inflow of water and electrolytes into the colonic lumen.
7) Insufficient water absorption in the small intestine following total or partial resection of the large intestine (ileostomy).
DiagnosisTop
History and Physical Examination
As there is a broad range of GI and non-GI conditions that can lead to diarrhea, it is difficult to advise a single diagnostic approach. A detailed history and physical examination are recommended to tailor investigations.Evidence 1Strong recommendation (benefits clearly outweigh downsides; right action for all or almost all patients; best practice statement). Arasaradnam RP, Brown S, Forbes A, et al. Guidelines for the investigation of chronic diarrhoea in adults: British Society of Gastroenterology, 3rd edition. Gut. 2018 Aug;67(8):1380-1399. doi: 10.1136/gutjnl-2017-315909. Epub 2018 Apr 13. PMID: 29653941; PMCID: PMC6204957. Review stool characteristics and frequency, associated symptoms (abdominal pain, stress, anxiety), sick contacts, diet, water source, medications, comorbidities (diabetes, thyroid disorders, HIV infection, immune disorders), and surgical history. Specifically inquire about alarm features including new symptoms after the age of 50, rectal bleeding, unintentional weight loss, iron deficiency anemia, nocturnal symptoms, and family history of colon cancer. Examine patients for signs of dehydration, nutritional status, extraintestinal manifestations of IBD, lymphadenopathy, and abdominal tenderness. Assess the severity of dehydration in every patient. Consider rectal examination to assess anal sphincter tone and to evaluate for the presence of perianal ulcers or fistulas.
Acute Diarrhea
If no characteristic manifestations are seen and the patient’s history does not suggest any noninfectious cause (eg, drugs), acute diarrhea is likely caused by a GI infection or food poisoning (see Acute Infectious Diarrhea). If symptoms persist or worsen despite appropriate treatment and diarrhea is ongoing for >10 days or diarrheal episodes recur frequently, consider causes of chronic diarrhea and perform appropriate workup (Figure 1.9-2).
Chronic Diarrhea
Characteristics of stools can be helpful in narrowing the differential diagnosis of possible causes. Initial screening blood tests are recommended concomitantly or consecutively, if the diagnosis is elusive, to investigate for organic disease and screen for malabsorptive conditions, including a complete blood count, electrolytes, liver function tests, vitamin B12, calcium, ferritin, C-reactive protein (CRP), and celiac disease screening (anti-tissue transglutaminase and quantitative immunoglobulins). Fecal calprotectin is recommended by some societies to exclude colonic inflammation, especially in individuals with suspected IBS aged <40 years.
Further investigations may include the following actions:
1) Consider testing stools for ova and parasites or fecal leukocytes; Clostridioides difficile toxin assay; and stool pH. Stool may be examined for fat (spot fecal fat, quantitative 3-day fecal fat, or fecal elastase; if steatorrhea is considered, 3-day fecal fat collection is usually performed).
2) Consider investigations for lactase deficiency (hydrogen breath tests or impact of a lactose-free diet if lactase deficiency is a possibility).
3) Depending on the suspected etiology and clinical course, investigate towards, for example, pancreatic insufficiency, bacterial overgrowth, or laxative abuse.
4) Exclude spurious diarrhea, which is frequent passage of small amounts of liquid brown stool, typically accompanied by fecal incontinence. It is related to overfilling and distention of the colon caused by fecal impaction in the rectum in patients with severe constipation or by narrowing of the sigmoid colon (less frequently of the rectum) due to organic causes, including tumors.
Gastroduodenoscopy, colonoscopy with ileal intubation, or both, with or without biopsy may be valuable in certain situations (see Figure 1.9-2).
Fecal osmotic gap is useful to distinguish between secretory and osmotic diarrhea; however, it is rarely used in clinical practice. It is expressed in mOsm/L and may be either measured (stool osmolality − 2 × [Nastool + Kstool]) or estimated (290 − 2 × [Nastool + Kstool]).
The following features of diarrhea may further assist in directing investigation and determining etiology:
1. Secretory diarrhea: Stools are typically abundant (up to several liters a day), watery, have a fecal sodium concentration >70 mmol/L and low osmotic gap (<50 mOsm/L). Abdominal pain is usually absent. Fasting does not reduce the frequency of bowel movements or stool volumes (diarrhea wakes the patient at night); exceptions include patients with a history of bowel resection and with intestinal fistulas (anatomic or functional short bowel syndrome) in whom diarrhea is exacerbated by oral or enteral nutrition.
2. Osmotic diarrhea: Stools are characterized by a high osmotic gap (>125 mOsm/L) and sodium concentration <70 mmol/L. Foamy and explosive stools with a pH <5.5 and fecal reducing substances >0.5% indicate disaccharide maldigestion. Osmotic diarrhea is stopped by fasting and by withholding ingestion of the osmotically active substance.
3. Steatorrhea: Stools are fatty, shiny, sticky, difficult to flush, and foul smelling. If steatorrhea is confirmed, further investigations towards pancreatic insufficiency may be needed.
4. Inflammatory diarrhea: This may be manifested by the presence of blood in stool, high fecal white blood cell count, or positive fecal lactoferrin/calprotectin test results. It may be accompanied by systemic manifestations of inflammation (fever, elevated acute-phase protein [eg, CRP] levels, elevated erythrocyte sedimentation rate) or peripheral eosinophilia (eg, in allergic diarrhea) and hypoalbuminemia.
TreatmentTop
1) Oral loperamide is an opioid derivative that decreases bowel peristalsis, increases water absorption, and decreases the number of bowel movements. It should be considered as supportive treatment in patients with watery diarrhea without fever or with mild fever. Loperamide is well tolerated in adolescents and adults and is associated with low risk of adverse reactions. Start from 4 mg followed by 2 mg after each diarrheal stool (up to 16 mg/d). Loperamide is contraindicated in patients with bloody diarrhea and in those with high-grade fever.
2) Diphenoxylate/atropine (trade name Lomotil; activator of opioid receptors): 5 mg qid until control is achieved (maximum dose of diphenoxylate, 20 mg/d), then reduce the dose as needed. Maintenance doses may be as low as 25% of the initial daily dose required for achieving control.
3) Oral bismuth: ~524 mg every 30 to 60 minutes or 1050 mg every 60 minutes as needed for up to 2 days (max, ~4200 mg [8 doses of regular strength or 4 doses of maximum strength] per 24 h).
4) Diosmectite is an intestinal content absorbent. The usual dose is 3 g orally bid to tid.
Identification of the underlying etiology is critical to tailor treatment choices.
1) IBS with predominant diarrhea: see IBS.
2) Bile acid diarrhea: Use bile acid sequestrants: cholestyramine (powder, initially 2-4 g/d in single or divided doses; titrate based on response to a max of 24 g/d), colestipol (2-g tablet once daily or bid; titrate to a max of 16 g), colesevelam (powder, 3.75 g/d or 3 tablets of 625 mg bid).
3) Pancreatic enzymes: A starting dose is 500 units of lipase/kg/meal (usually 25,000-40,000 units/meal). Titrate up to 2500 units of lipase/kg/meal. Viokase is an uncoated formulation that additionally requires acid suppression to prevent acid-induced deactivation of pancreatic enzymes, whereas Creon is enteric coated and acid suppression is not needed.
4) Octreotide is used for symptomatic treatment of diarrhea associated with chemotherapy, Zollinger-Ellison syndrome, carcinoid tumor, ileostomy, fistula, bowel obstruction, and for symptomatic treatment of chronic diarrhea in patients with AIDS. Octreotide reduces visceral blood flow, inhibits intestinal secretion, and normalizes intestinal peristalsis. It is administered as subcutaneous or intermittent injections, usually at a dose of 300 to 600 microg/d.
ComplicationsTop
Dehydration is the key complication of acute and chronic diarrhea. The severity of dehydration determines the techniques and intensity of rehydration therapy (oral or IV, outpatient or inpatient, volume of administered fluids). The most accurate assessment of the severity of dehydration is based on comparing the patient’s current body weight with the most recent weight before the onset of diarrhea. The severity of dehydration is expressed as a percentage of body weight loss, which determines the volume of fluid that needs to be administered during the rehydration phase. Dehydration can also be estimated on the basis of clinical manifestations and is classified approximately as:
1) No clinical manifestations of dehydration (loss of <3% of body weight): No signs or symptoms of dehydration.
2) Mild dehydration (loss of 3%-5% of body weight): Increased thirst (absent in elderly patients with impaired sensation of thirst) and dry oral mucosa.
3) Moderate dehydration (loss of >5% but <10% of body weight): Markedly increased thirst, dry oral mucosa, sunken eyes, oliguria, postural hypotension, prolonged capillary refill time (for >2 seconds apply pressure to a nail bed until it blanches, then release; the pink color normally reappears within <1.5 seconds of releasing pressure), delayed skin fold recoil (assessed on the abdomen) and dry skin (dry axilla increases the odds of moderate dehydration ~3-fold).
4) Severe dehydration (loss of ≥10% of body weight): Manifestations of moderate dehydration accompanied by features of hypovolemic shock.
1. Indications for hospitalization (IV rehydration necessary): Severe dehydration, manifestations of dehydration in an elderly patient (these patients often have no sensation of thirst and drink insufficient amounts of fluids), conditions precluding oral rehydration (persistent vomiting or adynamic ileus), and failure of oral rehydration (signs and symptoms of dehydration worsening despite receiving seemingly appropriate amounts of oral rehydration solutions [ORSs] or difficulty drinking the required amounts of liquids).
2. Rehydration therapy is the mainstay of symptomatic treatment of diarrhea. Most patients with mild to moderate dehydration (loss of <10% of body weight) may be managed using an outpatient or home oral rehydration therapy. It is usually sufficient to take regular fluids (water, juices, isotonic drinks, soups) and table salt (eg, salt crackers). For hydration in children and the elderly with severe diarrhea, commercial glucose-electrolyte ORSs are available. Frequent administration of small portions of cold (refrigerated) ORS improves the patient’s tolerance. Fluid therapy consists of 2 phases, deficit replacement and maintenance therapy.
Replacement of deficits (rehydration): For the first 3 to 4 hours, administer ORS only. The volume of ORS should be equal to the estimated weight loss (the patient should drink freely to quench thirst):
1) No signs of dehydration: Up to ~20 mL/kg.
2) Mild dehydration: ~40 mL/kg.
3) Moderate dehydration: ~70 mL/kg.
4) Compensate for ongoing fluid loss by adding 5 mL/kg to the calculated volume after each diarrheal stool or episode of vomiting.
Patients with severe dehydration, signs of shock, or evidence of bowel obstruction require immediate hospitalization and IV administration of crystalloids. After the patient’s condition has stabilized (normalization of vital signs, improved level of consciousness, resolution of signs of intestinal obstruction), replace the estimated fluid deficit. Depending on the degree of improvement, continue IV rehydration or switch to oral rehydration using ORS (see above). Frequent reassessment is crucial.
Maintenance therapy: Continue ORS to replace the water and electrolytes lost with stool and emesis (see above) and start oral nutrition (realimentation). In addition, the patient should drink ORS or neutral fluids (in unlimited amounts until thirst is quenched) ensuring that the total fluid intake meets the basic daily fluid requirement (after subtracting the volume of ingested foods). ORS should be administered until the resolution of diarrhea.
3. Management of other abnormalities: If necessary (usually in patients with severe dehydration), treat metabolic acidosis, hypernatremia, hyponatremia, hypokalemia, hypocalcemia, and hypomagnesemia (see Electrolyte, Fluid, and Acid-Base Balance Disorders). Isotonic dehydration is most frequently seen in patients with diarrhea. In patients with chronic diarrhea, also treat malnutrition, vitamin deficiencies, and trace element deficiencies.
FiguresTop
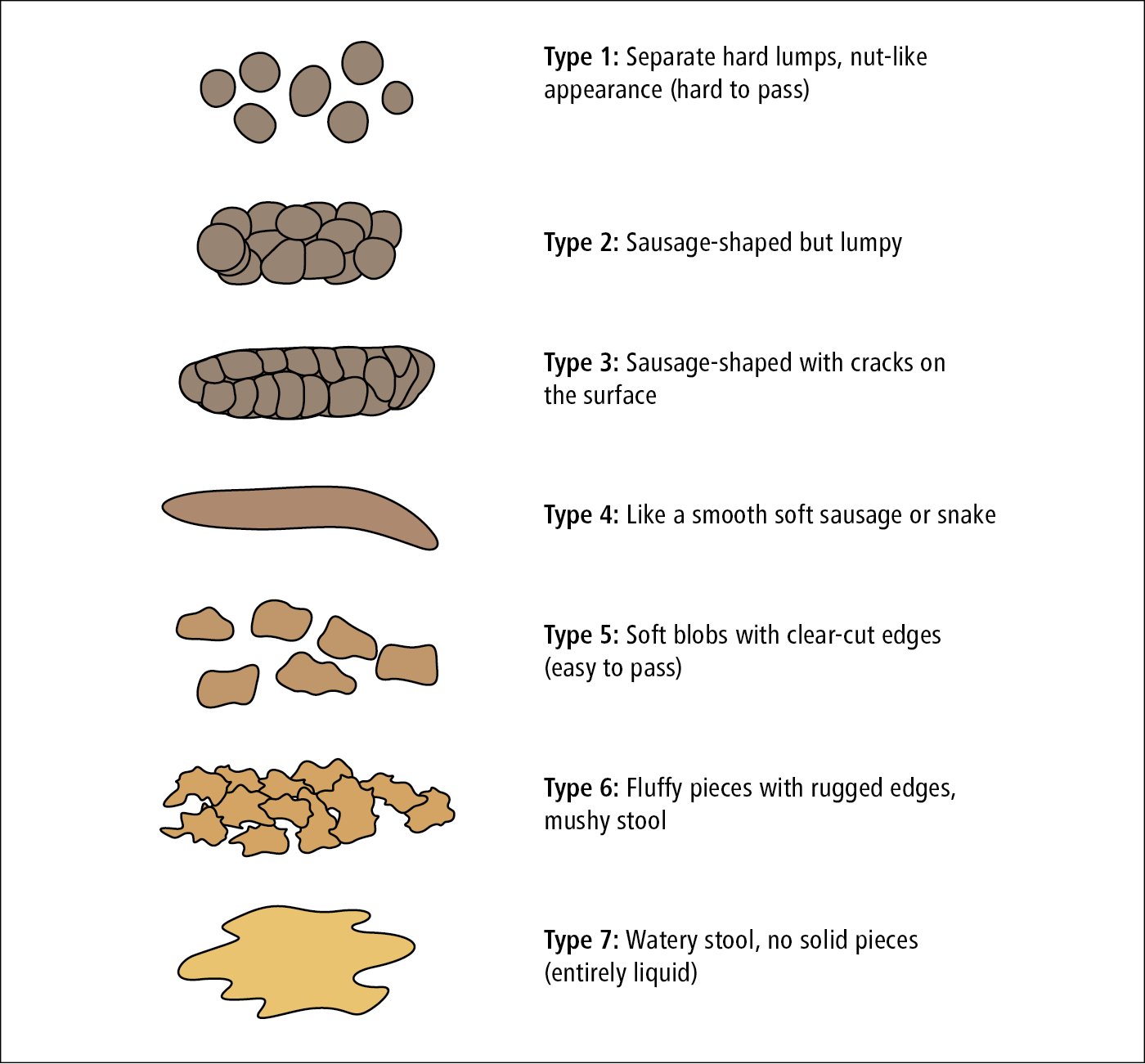
Figure 1.9-1. Bristol stool scale.
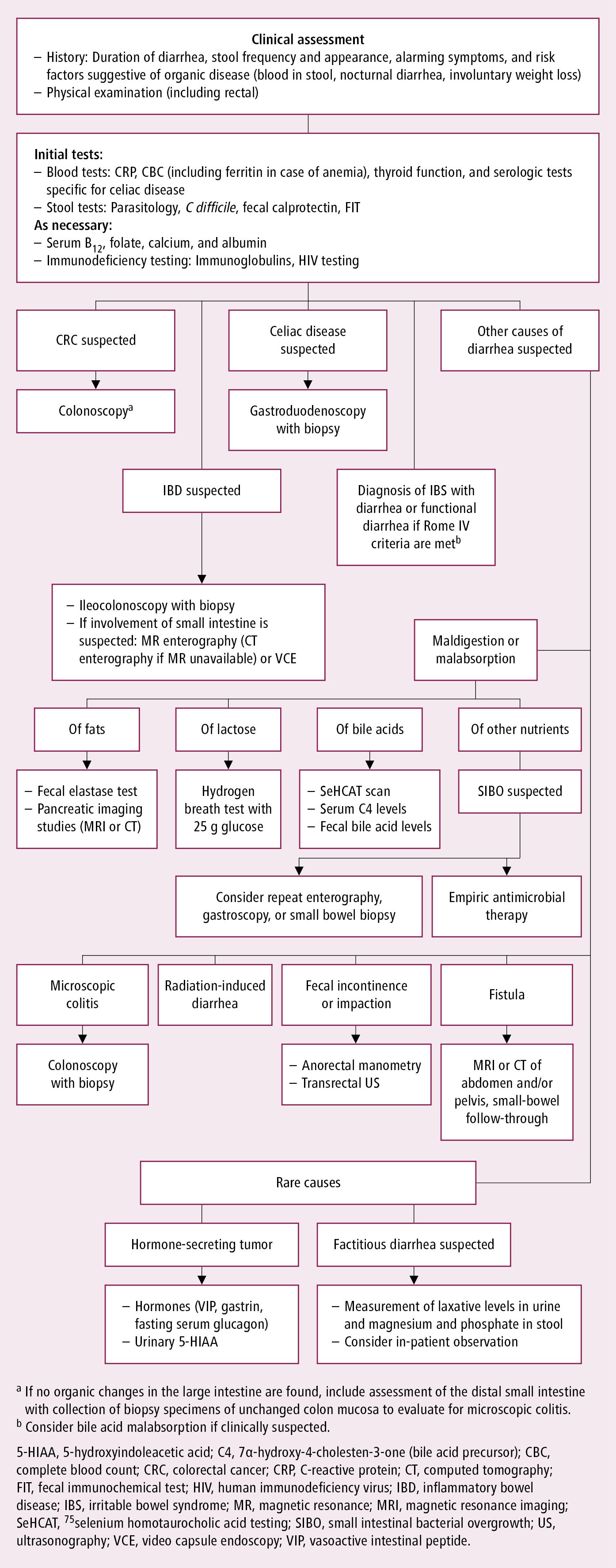
Figure 1.9-2. Algorithm for the diagnosis of chronic diarrhea. Adapted from Gut. 2018;67(8):1380-1399.